Region:Asia
Author(s):Dev
Product Code:KRAB1943
Pages:92
Published On:January 2026
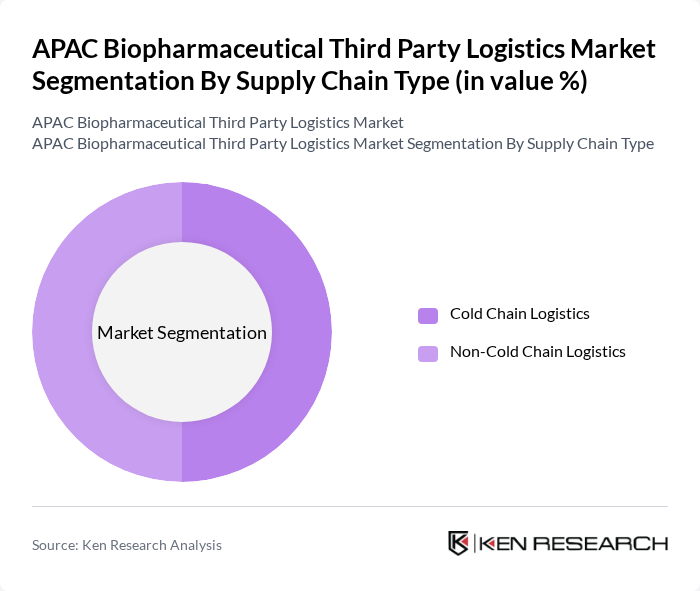
By Supply Chain Type:The supply chain type segmentation includes Cold Chain Logistics and Non-Cold Chain Logistics. Cold Chain Logistics is crucial for transporting temperature-sensitive biopharmaceuticals, including vaccines, biologics, and advanced therapies, ensuring product integrity and compliance with Good Distribution Practice and other regulatory standards. Non-Cold Chain Logistics, while also significant, primarily handles products that do not require temperature control and typically represents the larger share of overall pharmaceutical logistics volumes due to the high flow of conventional medicines. Globally, non-cold chain logistics has historically accounted for a higher share than cold chain in biopharmaceutical third party logistics, with cold chain expected to grow faster as demand for biologics and vaccines accelerates. Within APAC biopharmaceutical third party logistics, cold chain is expanding rapidly and its share is rising, though non-cold chain still holds a substantial proportion of the market.
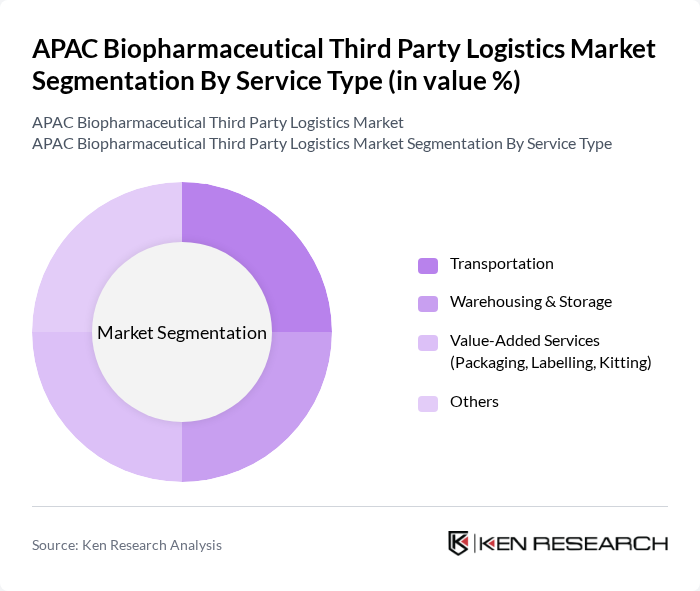
By Service Type:The service type segmentation encompasses Transportation, Warehousing & Storage, Value-Added Services (Packaging, Labelling, Kitting), and Others. Transportation is a critical service, as it directly impacts the timely and secure delivery of biopharmaceutical products to healthcare providers, distributors, and patients. Warehousing & Storage is also vital, providing GDP-compliant, temperature?controlled facilities for inventory management, consolidation, and quality assurance. Value-Added Services (such as secondary packaging, labelling, kitting, order fulfillment, and regulatory documentation support) are increasingly being adopted to enhance product handling, customization, and compliance and to enable integrated end?to?end solutions. At a global level, warehousing and storage has recently contributed the largest share of biopharmaceutical third party logistics revenue, while transportation remains the largest service category in the broader Asia Pacific pharmaceutical logistics market, indicating strong demand for both storage and transport services in APAC biopharma logistics.
The APAC Biopharmaceutical Third Party Logistics Market is characterized by a dynamic mix of regional and international players. Leading participants such as DHL Supply Chain, Kuehne + Nagel, DB Schenker, UPS Healthcare, FedEx, CEVA Logistics, GEODIS, Nippon Express, Kintetsu World Express, Yamato Transport, SF Express, Sinotrans, Kerry Logistics Network, CJ Logistics, Yusen Logistics contribute to innovation, geographic expansion, and service delivery in this space.
The APAC biopharmaceutical third-party logistics market is poised for significant transformation, driven by technological advancements and evolving consumer expectations. The integration of IoT and AI technologies is expected to enhance operational efficiency, enabling real-time monitoring and predictive analytics. Additionally, as sustainability becomes a priority, logistics providers are likely to adopt greener practices, reducing their carbon footprint. These trends will shape the future landscape, fostering innovation and improving service delivery in the biopharmaceutical logistics sector.
| Segment | Sub-Segments |
|---|---|
| By Supply Chain Type | Cold Chain Logistics Non-Cold Chain Logistics |
| By Service Type | Transportation Warehousing & Storage Value-Added Services (Packaging, Labelling, Kitting) Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) Contract Development and Manufacturing Organizations (CDMOs) Hospitals, Clinics, and Specialty Pharmacies Others |
| By Temperature Range | Ambient Refrigerated (2°C–8°C) Frozen (-20°C and below) Deep-Frozen / Ultra-Low (-80°C and below) |
| By Mode of Transport | Road Air Sea Rail |
| By Logistics Model | Third-Party Logistics (3PL) Fourth-Party Logistics (4PL) Integrated Logistics Others |
| By Country | China India Japan South Korea Australia & New Zealand Southeast Asia Rest of APAC |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cold Chain Logistics Providers | 100 | Logistics Managers, Cold Chain Specialists |
| Biopharmaceutical Manufacturing Facilities | 80 | Operations Managers, Quality Assurance Leads |
| Regulatory Compliance Experts | 60 | Regulatory Affairs Managers, Compliance Officers |
| Distribution Network Analysts | 70 | Supply Chain Analysts, Distribution Managers |
| Technology Providers for Logistics | 50 | Product Managers, Technology Solutions Architects |
The APAC Biopharmaceutical Third Party Logistics Market is valued at approximately USD 1314 billion, reflecting its significant share within the global biopharmaceutical logistics sector, driven by increasing demand for biopharmaceuticals and advancements in healthcare infrastructure.