Region:Global
Author(s):Rebecca
Product Code:KRAB1005
Pages:80
Published On:December 2025
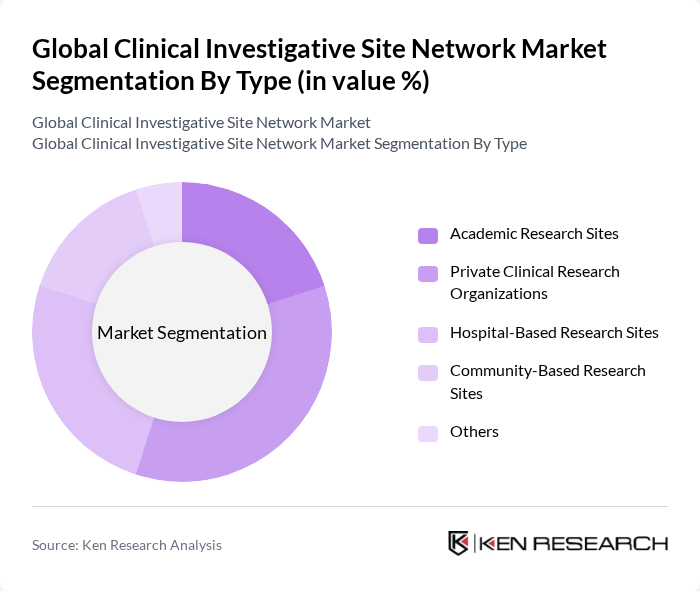
By Type:The market is segmented into various types, including Academic Research Sites, Private Clinical Research Organizations, Hospital-Based Research Sites, Community-Based Research Sites, and Others. Among these, Private Clinical Research Organizations are currently dominating the market due to their ability to provide specialized services and faster trial execution, which is increasingly sought after by pharmaceutical companies. The demand for efficient and compliant site management is driving growth in this segment.
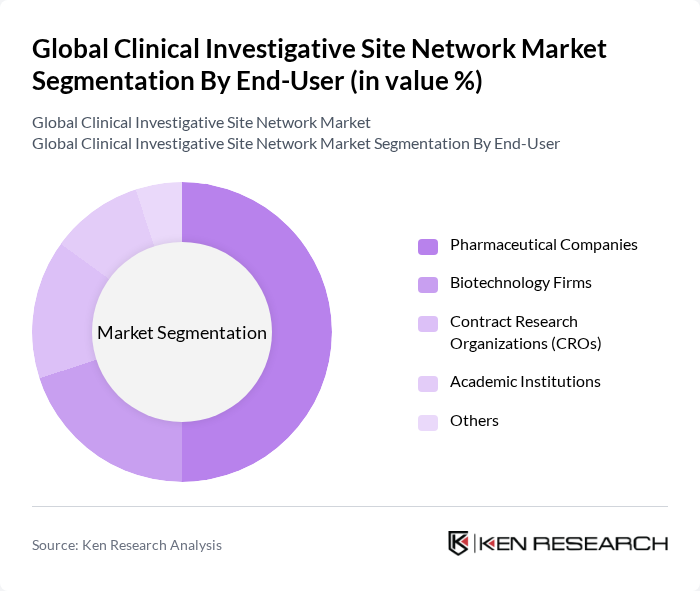
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Contract Research Organizations (CROs), Academic Institutions, and Others. Pharmaceutical Companies are the leading end-users, driven by their need for extensive clinical trials to bring new drugs to market. The increasing complexity of drug development and the need for specialized expertise in clinical trials are propelling the demand for services from this segment.
The Global Clinical Investigative Site Network Market is characterized by a dynamic mix of regional and international players. Leading participants such as Covance, PPD, ICON plc, Syneos Health, Medpace, Charles River Laboratories, Parexel International, WuXi AppTec, PRA Health Sciences, KCR, Clinipace, Worldwide Clinical Trials, InVentiv Health, Medidata Solutions, ERT contribute to innovation, geographic expansion, and service delivery in this space.
The future of the clinical investigative site network market appears promising, driven by a shift in trial volume towards emerging markets, particularly China, which is expected to surpass the U.S. in clinical trial listings. Additionally, the trend towards more transparent and cost-effective clinical trials, as demonstrated by recent initiatives, may reshape the operational landscape. These developments indicate a potential for increased collaboration and innovation within the industry, fostering a more dynamic research environment that prioritizes efficiency and patient engagement.
| Segment | Sub-Segments |
|---|---|
| By Type | Academic Research Sites Private Clinical Research Organizations Hospital-Based Research Sites Community-Based Research Sites Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) Academic Institutions Others |
| By Study Phase | Phase I Phase II Phase III Phase IV Others |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Others |
| By Geographic Focus | North America Europe Asia-Pacific Latin America Others |
| By Recruitment Strategy | Patient Recruitment Services Site Selection Services Retention Services Others |
| By Funding Source | Government Grants Private Investments Corporate Funding Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Clinical Trials | 100 | Clinical Research Coordinators, Oncologists |
| Cardiovascular Studies | 80 | Principal Investigators, Cardiologists |
| Neurology Research Initiatives | 70 | Clinical Trial Managers, Neurologists |
| Pediatric Clinical Trials | 60 | Pediatricians, Clinical Research Associates |
| Rare Disease Studies | 50 | Research Scientists, Regulatory Affairs Specialists |
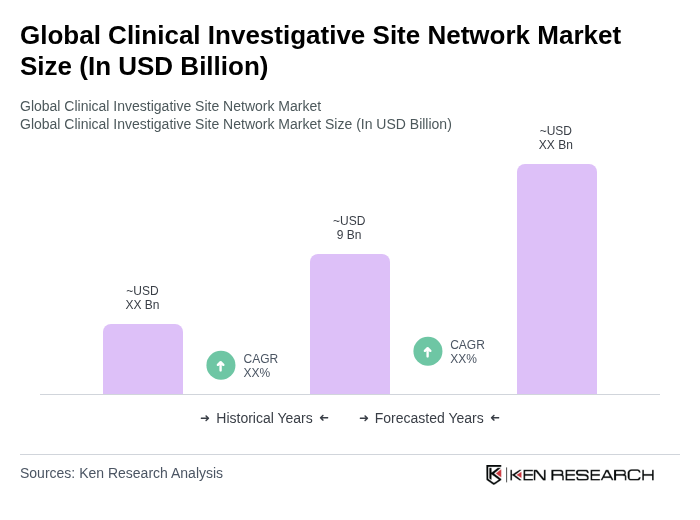
The Global Clinical Investigative Site Network Market is valued at approximately USD 9 billion, driven by increased investments in pharmaceutical R&D and a growing number of clinical trials across various therapeutic areas.