About the Report
Base Year 2024Indonesia Independent Diagnostic Testing Facility Market Overview
- The Indonesia Independent Diagnostic Testing Facility Market is valued at USD 2.4 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases, rising healthcare expenditure, advancements in diagnostic technologies, increasing healthcare awareness, and expansion of healthcare infrastructure in urban centers.
- Key cities such as Jakarta, Surabaya, and Bandung dominate the market due to their high population density and concentration of healthcare facilities. Jakarta, being the capital, has a robust healthcare infrastructure and a growing number of private diagnostic centers, while Surabaya and Bandung serve as significant regional hubs for healthcare services, attracting investments in diagnostic testing.
- The Minister of Health Regulation No. 65 of 2023 on Health Laboratory Standards issued by the Ministry of Health mandates that all independent diagnostic testing facilities must comply with national standards for operational scope, quality assurance, personnel qualifications, equipment calibration, and biosafety requirements.
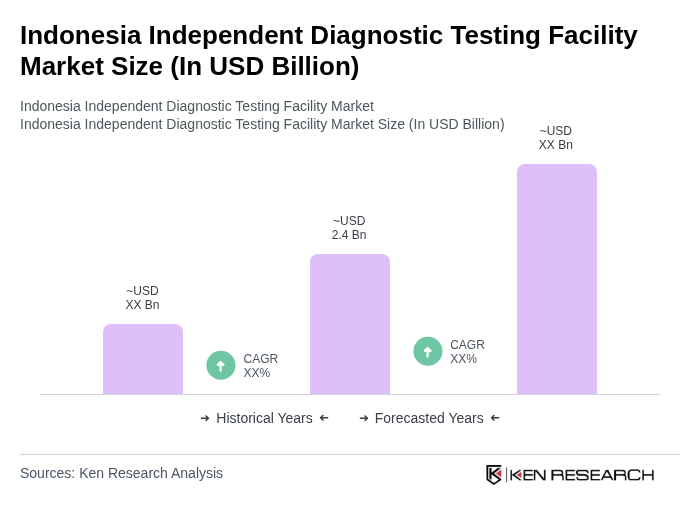
Indonesia Independent Diagnostic Testing Facility Market Segmentation
By Type:The market is segmented into various types of diagnostic testing, including Clinical Chemistry, Microbiology, Hematology, Immunology, Molecular Diagnostics, Pathology, and Others. Each of these segments plays a crucial role in the overall market dynamics, with specific applications and technologies driving their growth.
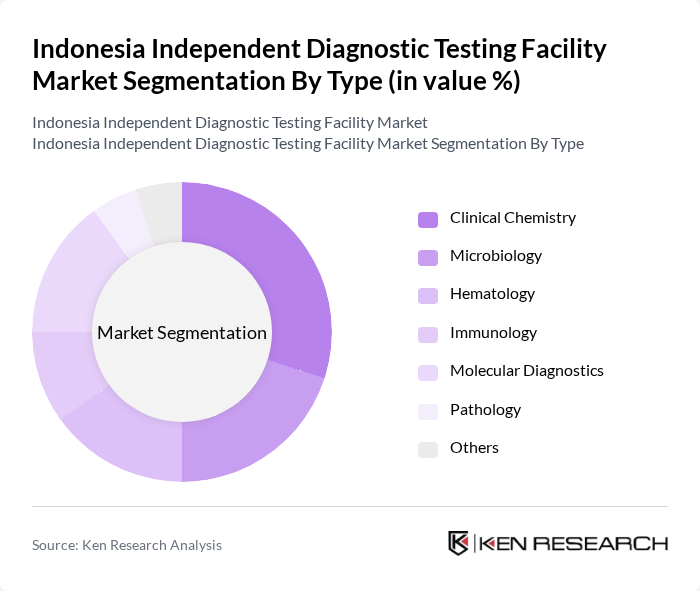
The Clinical Chemistry segment is currently dominating the market due to its extensive application in routine health check-ups and disease diagnosis. The increasing prevalence of metabolic disorders and the need for timely diagnosis have led to a surge in demand for clinical chemistry tests. Additionally, advancements in automated testing technologies have improved efficiency and accuracy, further driving the growth of this segment. The rising awareness among healthcare providers and patients about the importance of early diagnosis is also contributing to the segment's leadership.
By End-User:The market is segmented based on end-users, including Hospitals, Diagnostic Laboratories, Research Institutions, Home Care Settings, and Others. Each segment has unique requirements and contributes differently to the overall market landscape.
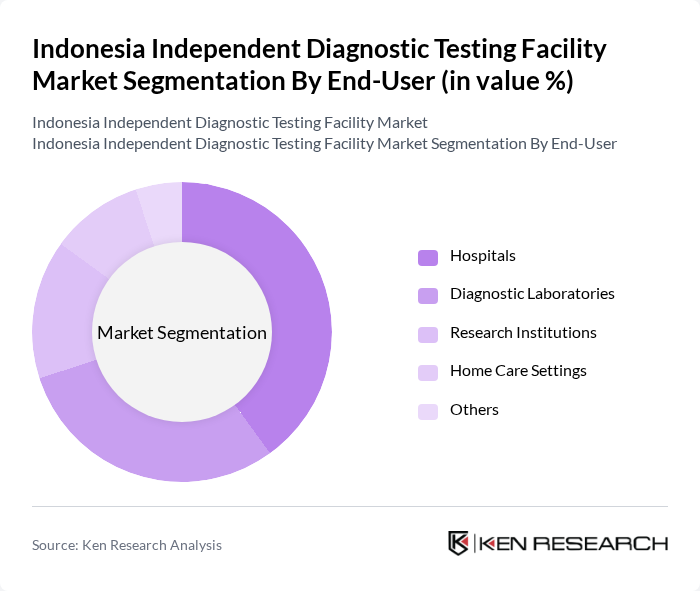
Hospitals are the leading end-user segment, accounting for a significant share of the market. This dominance is attributed to the high volume of diagnostic tests conducted in hospitals for patient management and treatment decisions. The integration of advanced diagnostic technologies in hospital settings has improved patient care and operational efficiency. Furthermore, the increasing number of hospitals and healthcare facilities in urban areas is expected to sustain the growth of this segment.
Indonesia Independent Diagnostic Testing Facility Market Competitive Landscape
The Indonesia Independent Diagnostic Testing Facility Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT. Prodia Widyahusada Tbk, PT. Kimia Farma Tbk, PT. Siloam International Hospitals Tbk, PT. Medikaloka Hermina Tbk, PT. Brawijaya Medika, PT. Bio Farma, PT. Indofarma Tbk, PT. Sumber Daya Manusia, PT. Citra Medika, PT. Sehat Selalu, PT. Medisafe Technologies, PT. Alkesindo, PT. Diagnos Medical, PT. Labora Medika, PT. Anugerah Medika contribute to innovation, geographic expansion, and service delivery in this space.
Indonesia Independent Diagnostic Testing Facility Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The rise in chronic diseases such as diabetes and cardiovascular conditions is a significant growth driver for the diagnostic testing market in Indonesia. According to the World Health Organization, approximately 11 million Indonesians are living with diabetes as of now. This increasing prevalence necessitates regular diagnostic testing, thereby boosting demand for independent diagnostic facilities. The healthcare expenditure in Indonesia is projected to reach IDR 1,400 trillion (approximately USD 93 billion) in future, further supporting this trend.
- Rising Demand for Early Diagnosis:The growing awareness of the importance of early diagnosis is propelling the independent diagnostic testing market. In future, around 65% of patients in urban areas sought early diagnostic services, reflecting a cultural shift towards preventive healthcare. The Indonesian government has allocated IDR 60 trillion (approximately USD 4 billion) for health initiatives in future, emphasizing early detection and treatment, which will likely enhance the market for diagnostic facilities significantly.
- Technological Advancements in Diagnostic Testing:Innovations in diagnostic technologies, such as rapid testing kits and AI-driven diagnostic tools, are transforming the landscape of independent diagnostic facilities. The market for diagnostic equipment in Indonesia is expected to grow to IDR 20 trillion (approximately USD 1.3 billion) by future, driven by these advancements. The integration of technology not only improves accuracy but also reduces turnaround times, making diagnostic services more appealing to healthcare providers and patients alike.
Market Challenges
- High Operational Costs:One of the primary challenges facing independent diagnostic testing facilities in Indonesia is the high operational costs associated with advanced diagnostic technologies and skilled labor. The average cost of setting up a diagnostic facility can exceed IDR 6 billion (approximately USD 400,000), which poses a barrier to entry for new players. Additionally, ongoing maintenance and staffing costs can strain financial resources, limiting the ability to compete effectively in the market.
- Regulatory Compliance Complexities:Navigating the regulatory landscape in Indonesia can be challenging for independent diagnostic facilities. The Ministry of Health has stringent regulations regarding laboratory accreditation and quality standards, which can be cumbersome and time-consuming. Facilities must comply with over 35 specific regulations, and failure to meet these standards can result in penalties or closure. This complexity can deter investment and slow down the establishment of new diagnostic facilities in the region.
Indonesia Independent Diagnostic Testing Facility Market Future Outlook
The future of the independent diagnostic testing facility market in Indonesia appears promising, driven by increasing healthcare investments and a focus on technological integration. As the government continues to enhance healthcare access, the demand for diagnostic services is expected to rise. Furthermore, the trend towards personalized medicine and telemedicine will likely create new avenues for growth, enabling facilities to offer tailored services that meet the evolving needs of patients and healthcare providers alike.
Market Opportunities
- Expansion of Telemedicine Services:The rise of telemedicine presents a significant opportunity for independent diagnostic facilities. With an estimated 35% of healthcare consultations in Indonesia expected to be conducted online in future, diagnostic facilities can partner with telehealth providers to offer remote testing services, enhancing patient access and convenience.
- Collaborations with Healthcare Providers:Forming strategic partnerships with hospitals and clinics can enhance service offerings for independent diagnostic facilities. Collaborations can lead to integrated care models, where diagnostic services are seamlessly incorporated into patient treatment plans, potentially increasing patient volume and improving overall healthcare outcomes in the region.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Clinical Chemistry Microbiology Hematology Immunology Molecular Diagnostics Pathology Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Home Care Settings Others |
| By Region | Java Sumatra Bali and Nusa Tenggara Kalimantan Sulawesi Maluku and Papua |
| By Technology | Automated Testing Systems Manual Testing Methods Point-of-Care Testing Devices Laboratory Information Management Systems Others |
| By Application | Infectious Disease Testing Cancer Screening Genetic Testing Cardiovascular Testing Others |
| By Investment Source | Private Investments Government Funding International Aid Public-Private Partnerships Others |
| By Policy Support | Subsidies for Diagnostic Facilities Tax Incentives for R&D Grants for Technology Adoption Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, National Agency of Drug and Food Control)
Healthcare Providers and Hospitals
Diagnostic Testing Equipment Manufacturers
Biotechnology Firms
Pharmaceutical Companies
Health Insurance Companies
Public Health Organizations
Players Mentioned in the Report:
PT. Prodia Widyahusada Tbk
PT. Kimia Farma Tbk
PT. Siloam International Hospitals Tbk
PT. Medikaloka Hermina Tbk
PT. Brawijaya Medika
PT. Bio Farma
PT. Indofarma Tbk
PT. Sumber Daya Manusia
PT. Citra Medika
PT. Sehat Selalu
PT. Medisafe Technologies
PT. Alkesindo
PT. Diagnos Medical
PT. Labora Medika
PT. Anugerah Medika
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia Independent Diagnostic Testing Facility Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia Independent Diagnostic Testing Facility Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia Independent Diagnostic Testing Facility Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Rising demand for early diagnosis
3.1.3 Technological advancements in diagnostic testing
3.1.4 Government initiatives to improve healthcare access
3.2 Market Challenges
3.2.1 High operational costs
3.2.2 Regulatory compliance complexities
3.2.3 Limited awareness among healthcare providers
3.2.4 Competition from established players
3.3 Market Opportunities
3.3.1 Expansion of telemedicine services
3.3.2 Collaborations with healthcare providers
3.3.3 Development of personalized medicine
3.3.4 Investment in research and development
3.4 Market Trends
3.4.1 Shift towards point-of-care testing
3.4.2 Integration of AI in diagnostic processes
3.4.3 Growing focus on preventive healthcare
3.4.4 Increasing patient-centric approaches
3.5 Government Regulation
3.5.1 Implementation of quality standards
3.5.2 Licensing requirements for diagnostic facilities
3.5.3 Regulations on data privacy and security
3.5.4 Guidelines for laboratory accreditation
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia Independent Diagnostic Testing Facility Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia Independent Diagnostic Testing Facility Market Segmentation
8.1 By Type
8.1.1 Clinical Chemistry
8.1.2 Microbiology
8.1.3 Hematology
8.1.4 Immunology
8.1.5 Molecular Diagnostics
8.1.6 Pathology
8.1.7 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Diagnostic Laboratories
8.2.3 Research Institutions
8.2.4 Home Care Settings
8.2.5 Others
8.3 By Region
8.3.1 Java
8.3.2 Sumatra
8.3.3 Bali and Nusa Tenggara
8.3.4 Kalimantan
8.3.5 Sulawesi
8.3.6 Maluku and Papua
8.4 By Technology
8.4.1 Automated Testing Systems
8.4.2 Manual Testing Methods
8.4.3 Point-of-Care Testing Devices
8.4.4 Laboratory Information Management Systems
8.4.5 Others
8.5 By Application
8.5.1 Infectious Disease Testing
8.5.2 Cancer Screening
8.5.3 Genetic Testing
8.5.4 Cardiovascular Testing
8.5.5 Others
8.6 By Investment Source
8.6.1 Private Investments
8.6.2 Government Funding
8.6.3 International Aid
8.6.4 Public-Private Partnerships
8.6.5 Others
8.7 By Policy Support
8.7.1 Subsidies for Diagnostic Facilities
8.7.2 Tax Incentives for R&D
8.7.3 Grants for Technology Adoption
8.7.4 Others
9. Indonesia Independent Diagnostic Testing Facility Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Average Test Turnaround Time
9.2.7 Pricing Strategy
9.2.8 Innovation Rate (New Tests Developed)
9.2.9 Compliance Rate with Regulatory Standards
9.2.10 Customer Satisfaction Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 PT. Prodia Widyahusada Tbk
9.5.2 PT. Kimia Farma Tbk
9.5.3 PT. Siloam International Hospitals Tbk
9.5.4 PT. Medikaloka Hermina Tbk
9.5.5 PT. Brawijaya Medika
9.5.6 PT. Bio Farma
9.5.7 PT. Indofarma Tbk
9.5.8 PT. Sumber Daya Manusia
9.5.9 PT. Citra Medika
9.5.10 PT. Sehat Selalu
9.5.11 PT. Medisafe Technologies
9.5.12 PT. Alkesindo
9.5.13 PT. Diagnos Medical
9.5.14 PT. Labora Medika
9.5.15 PT. Anugerah Medika
10. Indonesia Independent Diagnostic Testing Facility Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Social Affairs
10.1.4 Ministry of Defense
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Diagnostic Equipment Procurement
10.2.3 Technology Upgrades
10.2.4 Training and Development
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Diagnostic Laboratories
10.3.3 Research Institutions
10.3.4 Home Care Settings
10.4 User Readiness for Adoption
10.4.1 Awareness of Diagnostic Technologies
10.4.2 Training Needs
10.4.3 Infrastructure Readiness
10.4.4 Financial Readiness
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 ROI Measurement Techniques
10.5.2 Use Case Development
10.5.3 Long-term Sustainability Plans
10.5.4 Expansion into New Markets
11. Indonesia Independent Diagnostic Testing Facility Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging Strategies
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of government health reports and regulations related to diagnostic testing facilities
- Review of industry publications and market reports on the healthcare sector in Indonesia
- Examination of demographic and epidemiological data to identify testing needs
Primary Research
- Interviews with key stakeholders in the healthcare sector, including hospital administrators and laboratory managers
- Surveys conducted with healthcare professionals to assess the demand for diagnostic testing services
- Focus group discussions with patients to understand their experiences and expectations regarding diagnostic testing
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including government and private sector reports
- Triangulation of qualitative insights from interviews with quantitative data from surveys
- Sanity checks through expert panel reviews comprising industry veterans and healthcare analysts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on national healthcare expenditure and diagnostic testing trends
- Segmentation of the market by types of diagnostic tests and healthcare facilities
- Incorporation of growth rates from historical data and projected healthcare reforms
Bottom-up Modeling
- Collection of data on the number of diagnostic testing facilities and their operational capacities
- Estimation of average revenue per test based on pricing models from leading facilities
- Volume estimates based on patient demographics and disease prevalence rates
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating population growth, disease incidence, and healthcare access
- Scenario modeling based on potential regulatory changes and technological advancements in diagnostics
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Public Hospital Diagnostic Services | 100 | Hospital Administrators, Laboratory Directors |
| Private Diagnostic Laboratories | 80 | Operations Managers, Quality Assurance Officers |
| Healthcare Policy Makers | 50 | Government Health Officials, Regulatory Bodies |
| Patient Experience in Diagnostic Testing | 120 | Patients, Caregivers, Health Advocates |
| Emerging Technologies in Diagnostics | 70 | Healthcare Innovators, Technology Developers |
Frequently Asked Questions
What is the current value of the Indonesia Independent Diagnostic Testing Facility Market?
The Indonesia Independent Diagnostic Testing Facility Market is valued at approximately USD 2.4 billion, driven by factors such as the rising prevalence of chronic diseases, increased healthcare expenditure, and advancements in diagnostic technologies.