Japan Independent Diagnostic Testing Facility Market Overview
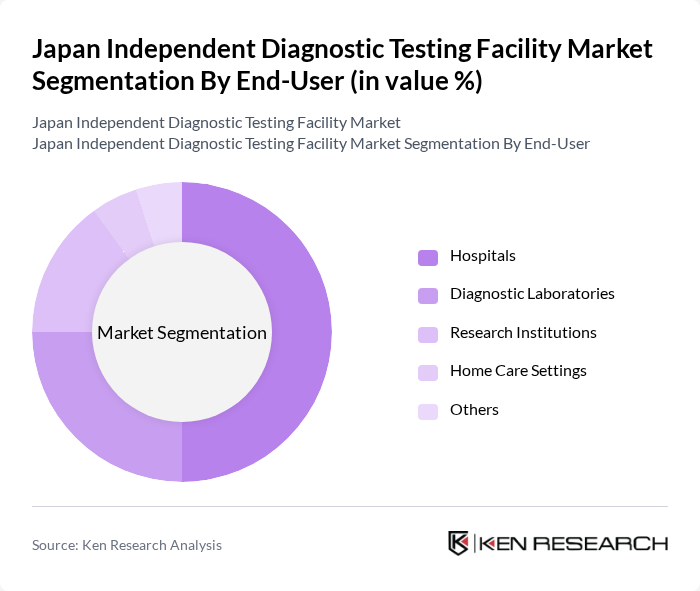
- The Japan Independent Diagnostic Testing Facility Market is valued at approximately USD 1.26 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases, advancements in diagnostic technologies, and a growing emphasis on preventive healthcare. The rise in healthcare expenditure and the demand for accurate and timely diagnostic services further contribute to the market's expansion. Japan's diagnostic utilization remains structurally high, with the country averaging more than 12 physician consultations per capita annually, significantly above OECD averages, reinforcing continuous laboratory demand.
- Tokyo, Osaka, and Yokohama are the dominant cities in the Japan Independent Diagnostic Testing Facility Market. Tokyo leads due to its status as a healthcare hub with numerous hospitals and research institutions. Osaka and Yokohama also play significant roles due to their advanced healthcare infrastructure and proximity to major pharmaceutical companies, fostering innovation and collaboration in diagnostic testing. The Kanto Region, which includes Tokyo, represents a major market concentration area alongside the Kansai/Kinki Region encompassing Osaka.
- The Japanese government has prioritized healthcare infrastructure investment through the Japan Agency for Medical Research and Development (AMED), funded by the Cabinet Secretariat, Ministry of Health, Labor and Welfare (MHLW), Ministry of Education, Culture, Sports, Science and Technology (MEXT), and Ministry of Economy, Trade and Industry (METI). In 2025, AMED allocated approximately USD 1.0 billion for research and development in medical technologies, health data, and diagnostics. Additionally, the MHLW increased its overall health budget by more than USD 3 billion in 2025, with portions allocated to strengthening regulatory capacity, medical device evaluation systems, and innovation support, improving the approval pipeline and commercialization environment for advanced diagnostic instruments.
Japan Independent Diagnostic Testing Facility Market Segmentation
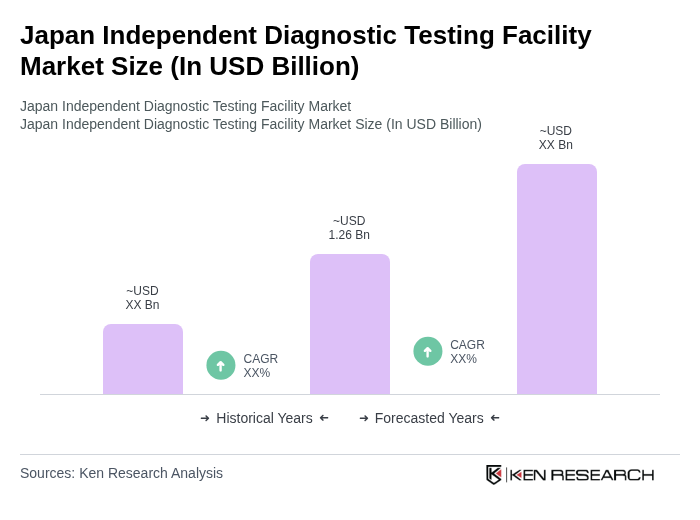
By Type:The market is segmented into various types, including Clinical Chemistry, Hematology, Microbiology, Immunology, Molecular Diagnostics, Pathology, and Others. Among these, Clinical Chemistry is the leading sub-segment, driven by the high demand for biochemical tests that are essential for diagnosing a wide range of diseases. The increasing prevalence of metabolic disorders and the need for routine health check-ups further bolster the growth of this segment.
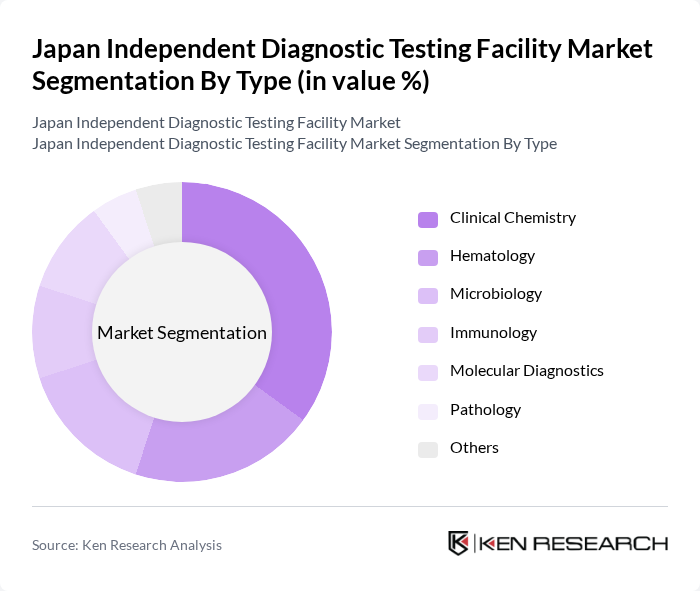
By End-User:The market is categorized by end-users, including Hospitals, Diagnostic Laboratories, Research Institutions, Home Care Settings, and Others. Hospitals represent the largest segment due to their comprehensive diagnostic services and the increasing number of patients requiring diagnostic tests. The growing trend of outpatient services and the need for rapid testing in emergency situations also contribute to the dominance of this segment. Japan has approximately 3,200 general hospitals, most operating in-house laboratories for routine testing, while outsourcing high-complexity molecular and genetic assays to centralized labs.
Japan Independent Diagnostic Testing Facility Market Competitive Landscape
The Japan Independent Diagnostic Testing Facility Market is characterized by a dynamic mix of regional and international players. Leading participants such as Fujirebio Inc., Sysmex Corporation, Roche Diagnostics K.K., Abbott Japan Co., Ltd., Siemens Healthineers K.K., Astellas Pharma Inc., Chugai Pharmaceutical Co., Ltd., Takeda Pharmaceutical Company Limited, Otsuka Pharmaceutical Co., Ltd., Mitsubishi Chemical Corporation, Medtronic Japan Co., Ltd., BML, Inc., Kanto Chemical Co., Inc., Nihon Kohden Corporation, Eiken Chemical Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space. Investment activity is concentrated in automation and centralization, with leading diagnostic groups investing heavily in mega-scale central laboratories equipped with total laboratory automation, robotics, and AI-enabled analyzers to process millions of samples annually.
Japan Independent Diagnostic Testing Facility Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The rise in chronic diseases such as diabetes and cardiovascular conditions is a significant growth driver for the independent diagnostic testing facility market in Japan. In future, approximately 12 million people are expected to be diagnosed with diabetes, reflecting a 20% increase from 2020. This growing patient population necessitates advanced diagnostic testing services, leading to increased demand for facilities that can provide timely and accurate results, thereby enhancing patient management and treatment outcomes.
- Rising Demand for Early Diagnosis:The emphasis on early diagnosis to improve treatment efficacy is propelling the independent diagnostic testing facility market. In future, Japan's healthcare system is projected to allocate ¥1.8 trillion (approximately $16.2 billion) towards early diagnostic technologies. This investment reflects a shift in healthcare strategies, focusing on preventive measures and early intervention, which is crucial for managing diseases effectively and reducing long-term healthcare costs.
- Technological Advancements in Diagnostic Testing:Continuous innovations in diagnostic technologies are transforming the landscape of independent testing facilities. In future, the market is expected to witness a 15% increase in the adoption of advanced diagnostic tools, including molecular diagnostics and next-generation sequencing. These technologies enhance accuracy and speed, allowing facilities to meet the growing demand for precise testing, ultimately improving patient outcomes and operational efficiency.
Market Challenges
- High Operational Costs:Independent diagnostic testing facilities face significant operational costs, which can hinder their growth. In future, the average operational cost for these facilities is projected to reach ¥600 million (approximately $5.4 million) annually. This includes expenses related to equipment maintenance, staffing, and compliance with regulatory standards. High costs can limit the ability of smaller facilities to compete effectively, impacting their market share and sustainability.
- Stringent Regulatory Requirements:The regulatory landscape for diagnostic testing in Japan is complex and stringent, posing challenges for independent facilities. Compliance with the Medical Device Regulation Act and guidelines from the Pharmaceutical and Medical Device Agency requires substantial investment in quality assurance and documentation processes. In future, facilities may incur costs exceeding ¥120 million (approximately $1.1 million) to meet these regulatory demands, which can strain financial resources and operational capabilities.
Japan Independent Diagnostic Testing Facility Market Future Outlook
The future of the independent diagnostic testing facility market in Japan appears promising, driven by technological advancements and a growing focus on preventive healthcare. As the healthcare landscape evolves, facilities are expected to increasingly adopt point-of-care testing and integrate artificial intelligence into diagnostic processes. This shift will enhance efficiency and accuracy, catering to the rising consumer demand for accessible and timely healthcare solutions. Additionally, the expansion of telehealth services will further facilitate remote diagnostics, broadening the reach of independent facilities.
Market Opportunities
- Expansion of Telehealth Services:The growing acceptance of telehealth presents a significant opportunity for independent diagnostic testing facilities. By integrating telehealth solutions, facilities can offer remote consultations and diagnostics, reaching a broader patient base. In future, the telehealth market in Japan is expected to exceed ¥400 billion (approximately $3.6 billion), indicating a substantial potential for collaboration and service expansion in the diagnostic sector.
- Development of Personalized Medicine:The shift towards personalized medicine is creating new avenues for independent diagnostic testing facilities. By focusing on tailored diagnostic solutions, facilities can enhance patient care and treatment outcomes. In future, investments in personalized medicine are projected to reach ¥250 billion (approximately $2.3 billion), highlighting the potential for facilities to innovate and differentiate their services in a competitive market.