Region:Asia
Author(s):Rebecca
Product Code:KRAA6084
Pages:96
Published On:January 2026
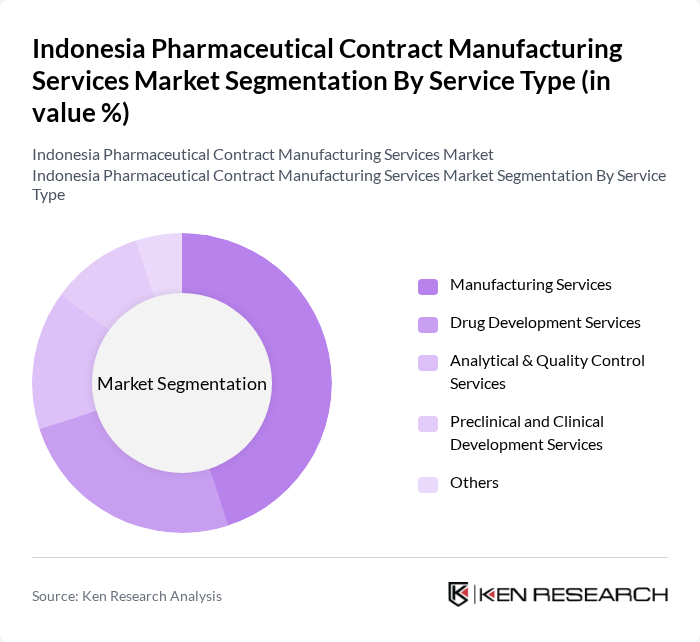
By Service Type:The service type segmentation includes various categories such as Manufacturing Services, Drug Development Services, Analytical & Quality Control Services, Preclinical and Clinical Development Services, and Others. Among these, Manufacturing Services is the leading segment due to the high demand for contract manufacturing as pharmaceutical companies seek to optimize production costs and focus on core competencies. The trend towards outsourcing manufacturing processes is expected to continue, driven by the need for efficiency and cost-effectiveness.
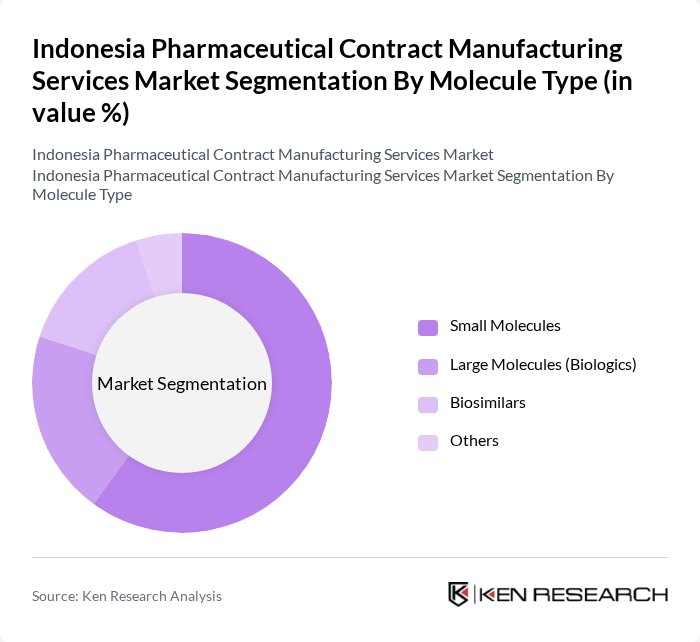
By Molecule Type:The molecule type segmentation encompasses Small Molecules, Large Molecules (Biologics), Biosimilars, and Others. The Small Molecules segment dominates the market, primarily due to their widespread use in various therapeutic areas and the established manufacturing processes associated with them. The increasing prevalence of chronic diseases and the demand for generic drugs further bolster the growth of this segment, making it a critical focus for contract manufacturers.
The Indonesia Pharmaceutical Contract Manufacturing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT Kimia Farma Tbk, PT Indofarma Tbk, PT Kalbe Farma Tbk, PT Dexa Medica, PT Sanbe Farma, PT Phapros Tbk, PT Merck Tbk, PT Sanofi Indonesia, PT Novartis Indonesia, PT Pfizer Indonesia, PT GlaxoSmithKline Indonesia, PT Johnson & Johnson Indonesia, PT Sido Muncul, PT Darya-Varia Laboratoria Tbk, PT Bio Farma contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Indonesian pharmaceutical contract manufacturing services market appears promising, driven by increasing healthcare demands and government support for local production. As the market evolves, advancements in technology and a focus on sustainable practices will likely shape the industry landscape. Additionally, the growing trend of outsourcing manufacturing will enable local firms to enhance their operational efficiencies and expand their service offerings, positioning them favorably in a competitive environment.
| Segment | Sub-Segments |
|---|---|
| By Service Type | Manufacturing Services Drug Development Services Analytical & Quality Control Services Preclinical and Clinical Development Services Others |
| By Molecule Type | Small Molecules Large Molecules (Biologics) Biosimilars Others |
| By Dosage Form | Tablets Capsules Sterile Injectables Liquids and Suspensions Topicals Others |
| By End-User | Pharmaceutical Companies (Innovators & Generics) Biotechnology Firms Research Institutions Virtual Pharma Companies Others |
| By Region | Java Sumatra Bali Kalimantan Sulawesi Others |
| By Manufacturing Process | Batch Manufacturing Continuous Manufacturing Others |
| By Regulatory Compliance Level | GMP Certified Non-GMP Certified Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Contract Manufacturers | 120 | Operations Managers, Production Supervisors |
| Pharmaceutical Companies Utilizing Contract Services | 100 | Procurement Directors, Product Managers |
| Regulatory Affairs Professionals | 60 | Compliance Officers, Quality Control Managers |
| Industry Experts and Analysts | 40 | Market Analysts, Industry Consultants |
| Healthcare Providers and Pharmacists | 80 | Pharmacists, Healthcare Administrators |
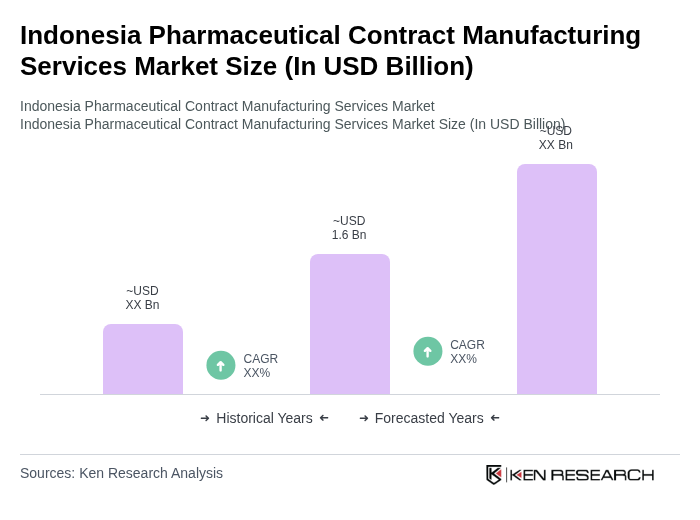
The Indonesia Pharmaceutical Contract Manufacturing Services Market is valued at approximately USD 1.6 billion, reflecting significant growth driven by increasing pharmaceutical demand, healthcare expenditure, and a focus on cost-effective manufacturing solutions.