Region:Asia
Author(s):Geetanshi
Product Code:KRVN7324
Pages:90
Published On:December 2025
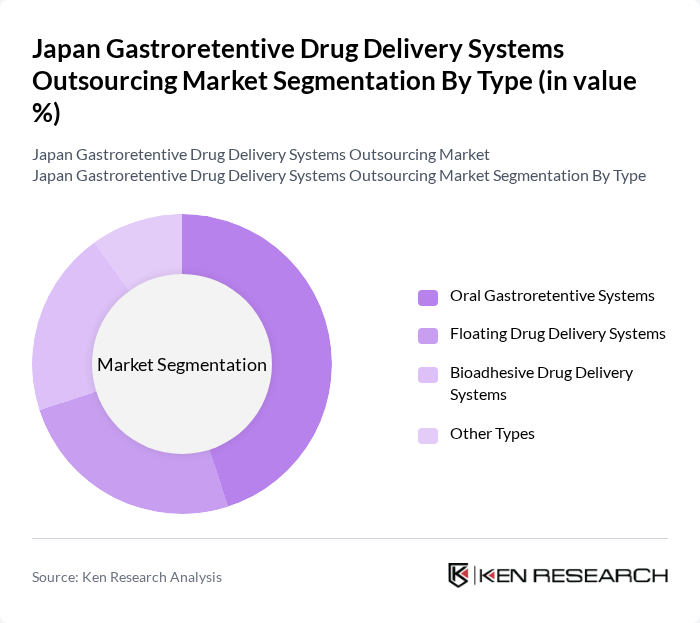
By Type:The market is segmented into various types of gastroretentive drug delivery systems, including Oral Gastroretentive Systems, Floating Drug Delivery Systems, Bioadhesive Drug Delivery Systems, and Other Types. Among these, Oral Gastroretentive Systems are leading due to their widespread acceptance and effectiveness in prolonging drug release and enhancing bioavailability. The growing preference for oral dosage forms among patients and healthcare providers further supports this trend.
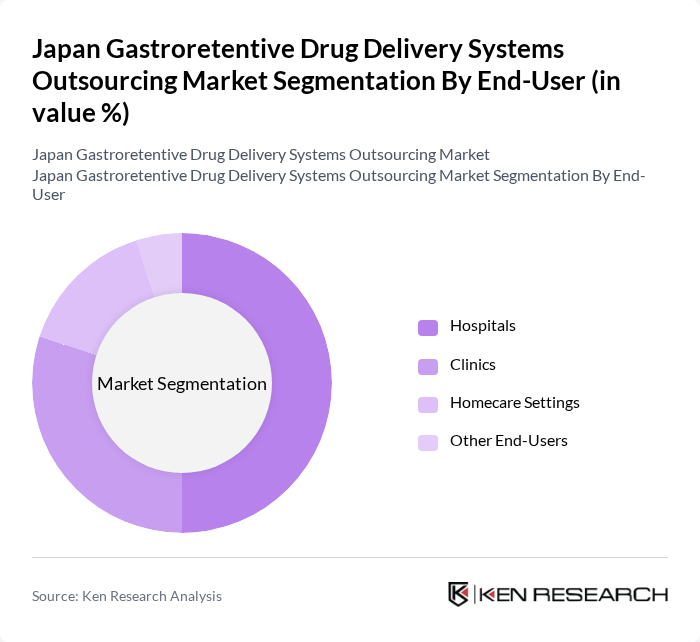
By End-User:The end-user segmentation includes Hospitals, Clinics, Homecare Settings, and Other End-Users. Hospitals are the dominant end-user segment, driven by the increasing number of surgical procedures and the need for effective postoperative care. The integration of advanced drug delivery systems in hospital settings enhances patient outcomes and reduces the length of hospital stays, making them a preferred choice for healthcare providers.
The Japan Gastroretentive Drug Delivery Systems Outsourcing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Astellas Pharma Inc., Takeda Pharmaceutical Company Limited, Daiichi Sankyo Company, Limited, Otsuka Pharmaceutical Co., Ltd., Chugai Pharmaceutical Co., Ltd., Mitsubishi Tanabe Pharma Corporation, Eisai Co., Ltd., Kyowa Kirin Co., Ltd., Shionogi & Co., Ltd., Sumitomo Dainippon Pharma Co., Ltd., Santen Pharmaceutical Co., Ltd., Kissei Pharmaceutical Co., Ltd., Astellas Gene Therapies, Maruho Co., Ltd., Medipal Holdings Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the gastroretentive drug delivery systems market in Japan appears promising, driven by ongoing advancements in technology and increasing healthcare demands. As the population ages, the prevalence of chronic gastrointestinal disorders is expected to rise, necessitating innovative drug delivery solutions. Furthermore, the integration of digital health technologies and patient-centric approaches will likely enhance treatment adherence. Collaborations between pharmaceutical companies and research institutions will also foster innovation, paving the way for new product developments and improved patient outcomes in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Oral Gastroretentive Systems Floating Drug Delivery Systems Bioadhesive Drug Delivery Systems Other Types |
| By End-User | Hospitals Clinics Homecare Settings Other End-Users |
| By Therapeutic Area | Gastroenterology Oncology Cardiovascular Other Therapeutic Areas |
| By Distribution Channel | Direct Sales Online Pharmacies Retail Pharmacies Other Channels |
| By Region | Kanto Kansai Chubu Other Regions |
| By Formulation | Tablets Capsules Suspensions Other Formulations |
| By Others | Custom Formulations Combination Therapies Other Innovations |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 100 | Product Development Managers, Regulatory Affairs Specialists |
| Healthcare Providers | 80 | Pharmacists, Physicians, Clinical Researchers |
| Research Institutions | 60 | Academic Researchers, Lab Directors |
| Regulatory Bodies | 50 | Policy Makers, Compliance Officers |
| Market Analysts | 70 | Market Research Analysts, Industry Consultants |
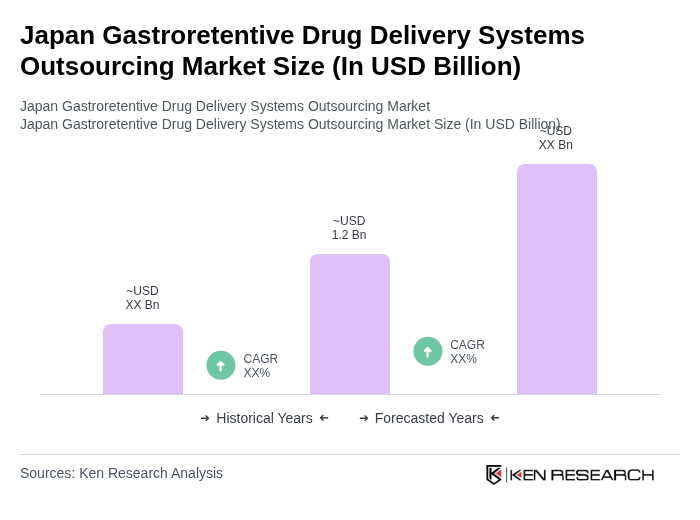
The Japan Gastroretentive Drug Delivery Systems Outsourcing Market is valued at approximately USD 1.2 billion, reflecting a significant growth driven by the increasing prevalence of gastrointestinal disorders and advancements in drug delivery technologies.