Region:Asia
Author(s):Dev
Product Code:KRAC5070
Pages:99
Published On:January 2026
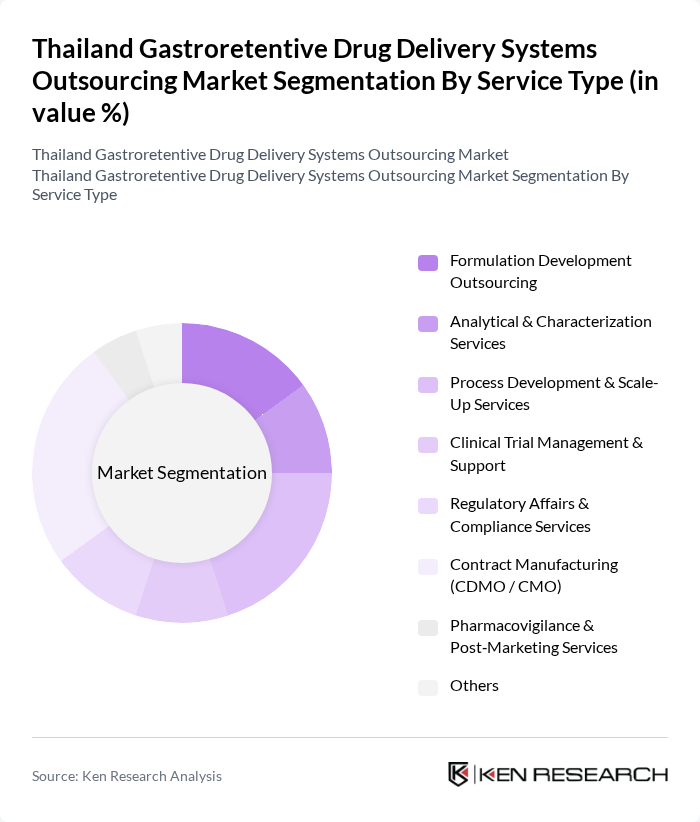
By Service Type:The service type segmentation includes various specialized services that cater to the needs of pharmaceutical companies in the development of gastroretentive drug delivery systems. The dominant sub-segment is Contract Manufacturing (CDMO / CMO), which is favored for its ability to provide comprehensive manufacturing solutions, allowing companies to focus on core competencies while ensuring high-quality production. This trend is driven by the increasing complexity of gastroretentive formulations (such as floating, expandable, and high?density systems), the need for specialized equipment for controlled?release tablet manufacturing, and the preference for partners with proven expertise in scaling up solid oral dosage forms that achieve prolonged gastric retention.
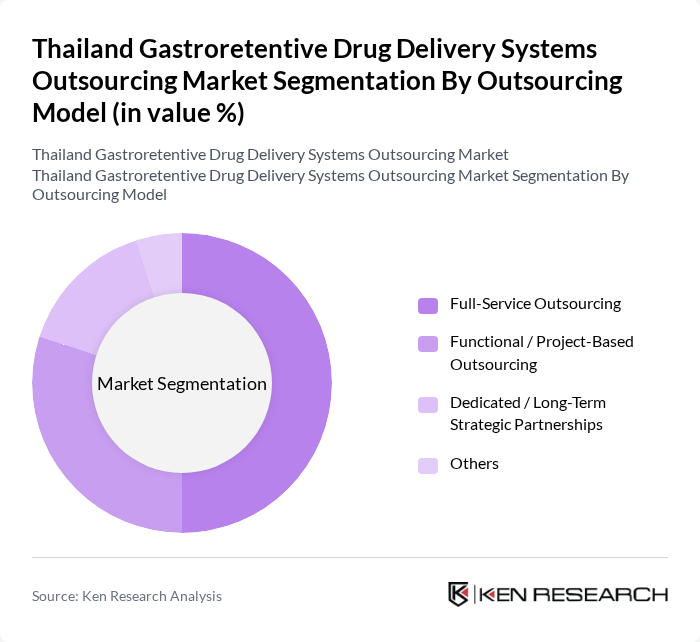
By Outsourcing Model:This segmentation focuses on the different models adopted by companies for outsourcing their drug delivery system development. The leading model is Full-Service Outsourcing, which allows pharmaceutical companies to engage a single partner for all aspects of drug development, from formulation to manufacturing. This model is preferred due to its efficiency, integrated quality management, and the ability to streamline processes across development, scale?up, and commercial manufacturing, thereby reducing time and costs associated with multiple vendor management and facilitating faster alignment with regulatory expectations for complex gastroretentive products.
The Thailand Gastroretentive Drug Delivery Systems Outsourcing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Siam Pharmaceutical Co., Ltd., The Government Pharmaceutical Organization (GPO), Thai Nakorn Patana Co., Ltd., Biolab Co., Ltd., Atlantic Pharmaceutical Co., Ltd., Greater Pharma Co., Ltd., Unison Laboratories Co., Ltd., T.O. Chemicals (1979) Co., Ltd., General Drugs House Co., Ltd., Sriprasit Pharma Co., Ltd., Biopharm Chemical Co., Ltd., Mega Lifesciences Public Company Limited, Bangkok Lab & Cosmetic Co., Ltd., Inno BioCosmed Co., Ltd., Bangkok Hospital Group (Bangkok Dusit Medical Services PCL) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the gastroretentive drug delivery systems outsourcing market in Thailand appears promising, driven by technological advancements and a shift towards patient-centric solutions. As healthcare infrastructure expands, the integration of digital health technologies will enhance drug delivery efficiency. Furthermore, the increasing focus on personalized medicine will likely lead to tailored gastroretentive systems, improving patient outcomes. These trends indicate a dynamic market landscape, fostering innovation and collaboration among stakeholders in the pharmaceutical sector.
| Segment | Sub-Segments |
|---|---|
| By Service Type | Formulation Development Outsourcing Analytical & Characterization Services Process Development & Scale-Up Services Clinical Trial Management & Support Regulatory Affairs & Compliance Services Contract Manufacturing (CDMO / CMO) Pharmacovigilance & Post?Marketing Services Others |
| By Outsourcing Model | Full-Service Outsourcing Functional / Project-Based Outsourcing Dedicated / Long-Term Strategic Partnerships Others |
| By Client Type | Multinational Pharmaceutical Companies Domestic Pharmaceutical Companies Biotechnology Firms Generic Drug Manufacturers Academic & Research Institutes Others |
| By Therapeutic Area | Gastrointestinal Disorders (e.g., ulcers, gastritis) Helicobacter pylori Infection Diabetes & Metabolic Disorders Cardiovascular Diseases Infectious Diseases Oncology Others |
| By Dosage Form / Technology | Floating Drug Delivery Systems Mucoadhesive Systems Swellable / Expandable Systems High?Density Systems Layered Tablets & Multi?Particulate Systems Capsules Others |
| By Stage of Development | Pre?Clinical Phase I Phase II Phase III Commercial / Post?Marketing |
| By Region | Central Thailand Northern Thailand Southern Thailand Northeastern Thailand |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 120 | Production Managers, Quality Assurance Heads |
| Healthcare Providers | 100 | Gastroenterologists, Clinical Pharmacists |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Research Institutions | 70 | Pharmaceutical Researchers, Academic Professors |
| Market Analysts | 60 | Market Research Analysts, Industry Consultants |
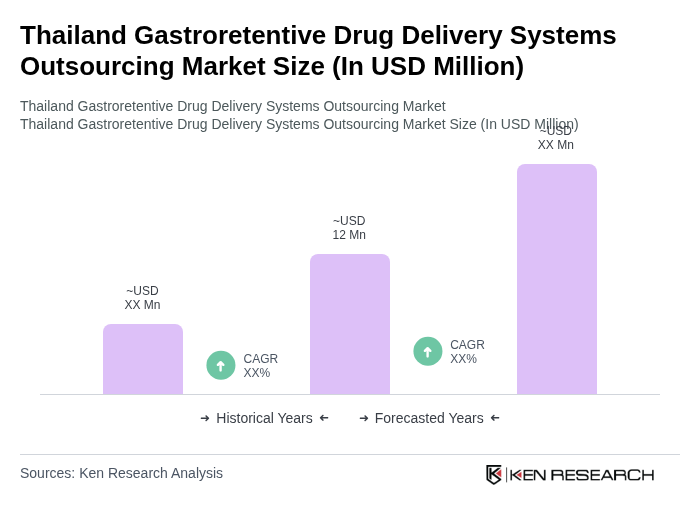
The Thailand Gastroretentive Drug Delivery Systems Outsourcing Market is valued at approximately USD 12 million, reflecting a five-year historical analysis that highlights growth driven by increasing gastrointestinal disorders and demand for innovative drug delivery systems.