Region:Africa
Author(s):Geetanshi
Product Code:KRVN7325
Pages:113
Published On:December 2025
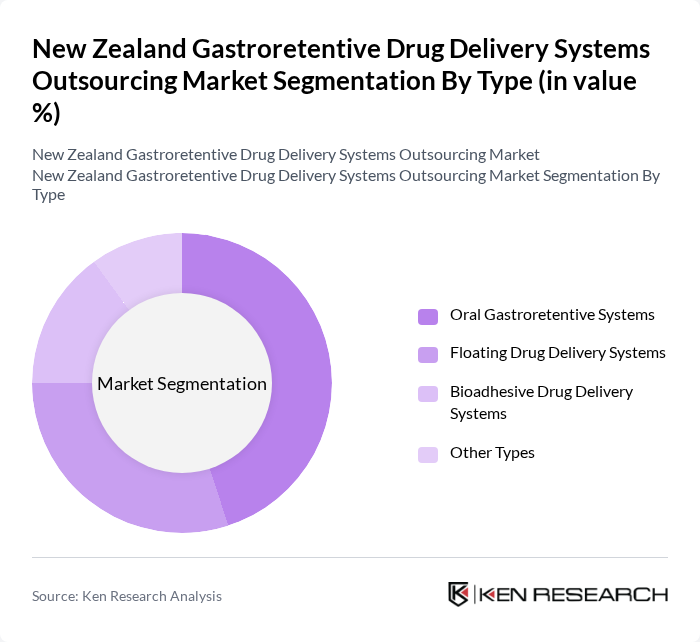
By Type:The market is segmented into various types of gastroretentive drug delivery systems, including oral gastroretentive systems, floating drug delivery systems, bioadhesive drug delivery systems, and other types. Among these, oral gastroretentive systems are leading the market due to their widespread acceptance and effectiveness in enhancing drug bioavailability. The demand for these systems is driven by their ability to provide sustained release of medications, improving patient adherence and therapeutic outcomes.
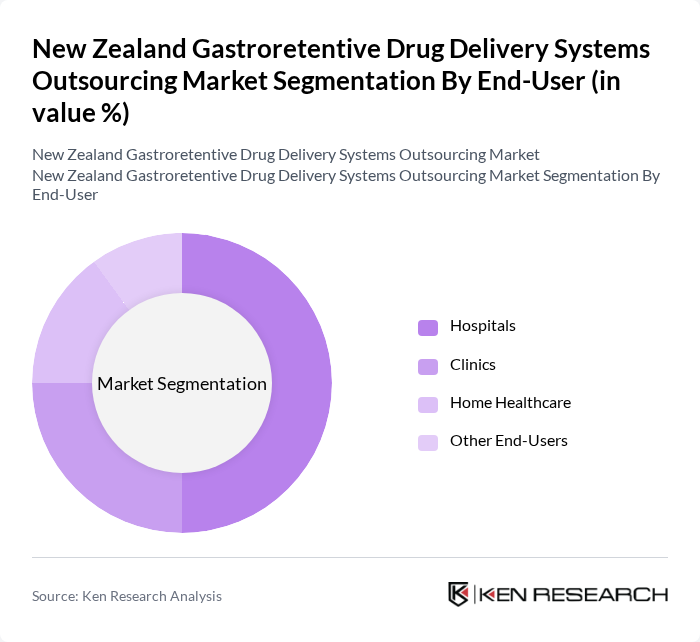
By End-User:The end-user segmentation includes hospitals, clinics, home healthcare, and other end-users. Hospitals are the dominant end-user segment, primarily due to their extensive patient base and the need for advanced drug delivery systems to manage complex medical conditions. The increasing adoption of gastroretentive systems in hospital settings is driven by their ability to improve drug efficacy and patient outcomes.
The New Zealand Gastroretentive Drug Delivery Systems Outsourcing Market is characterized by a dynamic mix of regional and international players. Leading participants such as AFT Pharmaceuticals, Douglas Pharmaceuticals, Pharmazen, Medsafe, Phebra, NZ Pharmaceuticals, Mylan, Teva Pharmaceuticals, Novartis, Pfizer, GSK, Merck, Sanofi, AstraZeneca, and Bayer contribute to innovation, geographic expansion, and service delivery in this space.
The future of the gastroretentive drug delivery systems outsourcing market in New Zealand appears promising, driven by increasing healthcare investments and a focus on innovative drug delivery solutions. As the healthcare sector continues to evolve, the integration of digital health technologies and personalized medicine will likely reshape the landscape. Companies that adapt to these trends and invest in research and development will be well-positioned to capitalize on emerging opportunities, ensuring sustainable growth in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Oral Gastroretentive Systems Floating Drug Delivery Systems Bioadhesive Drug Delivery Systems Other Types |
| By End-User | Hospitals Clinics Home Healthcare Other End-Users |
| By Application | Cardiovascular Diseases Diabetes Management Pain Management Other Applications |
| By Distribution Channel | Online Pharmacies Retail Pharmacies Hospital Pharmacies Other Channels |
| By Region | North Island South Island Other Regions |
| By Technology | Polymer-based Systems Gelatin-based Systems Other Technologies |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Other Policy Supports |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical R&D Departments | 100 | R&D Managers, Formulation Scientists |
| Clinical Research Organizations | 80 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Healthcare Providers and Hospitals | 70 | Pharmacists, Hospital Administrators |
| Regulatory Bodies | 50 | Regulatory Affairs Officers, Compliance Managers |
| Market Research Firms | 60 | Market Analysts, Industry Consultants |
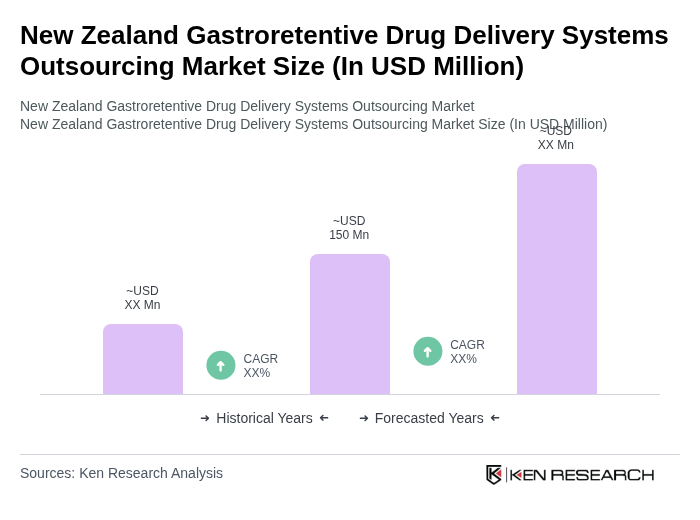
The New Zealand Gastroretentive Drug Delivery Systems Outsourcing Market is valued at approximately USD 150 million, reflecting a five-year historical analysis that highlights growth driven by chronic disease prevalence and advancements in drug delivery technologies.