Japan Pediatric Drugs Market Overview
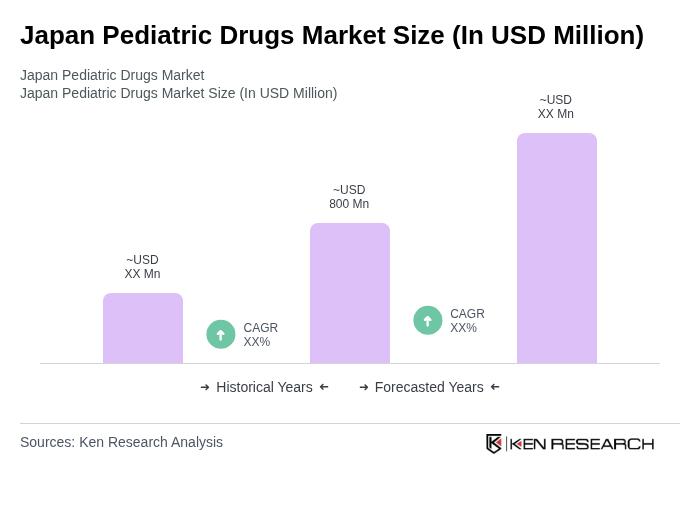
- The Japan Pediatric Drugs Market is valued at USD 800 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of pediatric diseases, advancements in drug formulations tailored for children, a growing focus on childhood chronic conditions and mental health concerns, and a growing awareness among parents regarding child health. The market is also supported by the rising number of pediatric healthcare facilities, government initiatives to improve child healthcare access, and the introduction of innovative therapies.
- Key cities such as Tokyo, Osaka, and Yokohama dominate the market due to their advanced healthcare infrastructure, high population density, and concentration of leading pharmaceutical companies. These urban centers are hubs for medical research and development, facilitating the rapid introduction of new pediatric drugs and therapies, thus enhancing market growth.
- The Ministerial Ordinance on Approval Standards for New Drugs, 2005 issued by Japan's Ministry of Health, Labour and Welfare requires pediatric drug applications to include data on pediatric pharmacokinetics, efficacy, and safety where pediatric use is anticipated, with compliance mandating age-appropriate formulations, dosing thresholds based on body weight or surface area, and post-marketing pediatric surveillance for drugs used in children under 15 years.
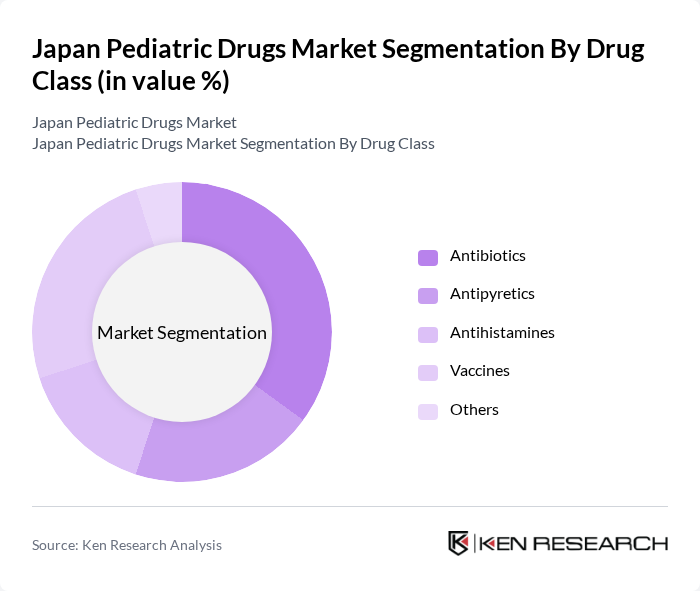
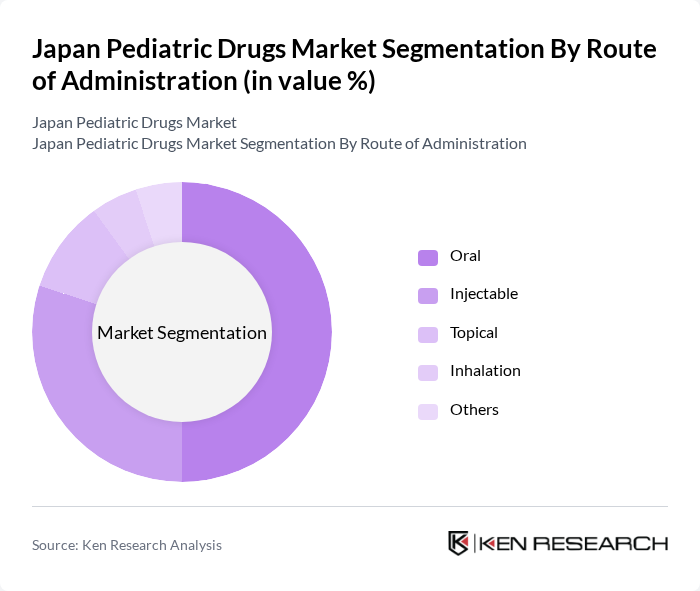
Japan Pediatric Drugs Market Segmentation
By Drug Class:The market is segmented into various drug classes, including antibiotics, antipyretics, antihistamines, vaccines, and others. Among these, antibiotics and vaccines are particularly significant due to their essential roles in treating infections and preventing diseases in children. The increasing incidence of infectious diseases and the growing vaccination programs contribute to the dominance of these segments.
By Route of Administration:The market is categorized based on the route of administration, which includes oral, injectable, topical, inhalation, and others. Oral administration is the most preferred method due to its ease of use and acceptance among children. Injectable forms are also significant, especially for vaccines and certain antibiotics, reflecting the diverse needs of pediatric patients.
Japan Pediatric Drugs Market Competitive Landscape
The Japan Pediatric Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Takeda Pharmaceutical Company Limited, Astellas Pharma Inc., Chugai Pharmaceutical Co., Ltd., Daiichi Sankyo Company, Limited, Eisai Co., Ltd., Otsuka Pharmaceutical Co., Ltd., Mitsubishi Tanabe Pharma Corporation, Shionogi & Co., Ltd., Kyowa Kirin Co., Ltd., Sumitomo Dainippon Pharma Co., Ltd., Johnson & Johnson, Pfizer Inc., Novartis AG, Merck & Co., Inc., Sanofi S.A. contribute to innovation, geographic expansion, and service delivery in this space.
Japan Pediatric Drugs Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Pediatric Diseases:The rise in pediatric diseases, such as asthma and diabetes, is a significant growth driver for the Japan pediatric drugs market. In future, approximately 1.3 million children in Japan are expected to be diagnosed with asthma, reflecting a 8% increase from 2020. Additionally, the prevalence of type 1 diabetes among children has risen to 0.3% of the pediatric population, necessitating effective drug formulations. This growing disease burden underscores the urgent need for innovative pediatric treatments.
- Rising Healthcare Expenditure:Japan's healthcare expenditure is projected to reach ¥50 trillion (approximately $450 billion) in future, driven by an aging population and increased focus on healthcare quality. This investment translates into enhanced access to pediatric drugs, as hospitals and clinics expand their offerings. The government’s commitment to improving healthcare infrastructure, including pediatric care, is expected to facilitate the development and distribution of essential medications for children, thereby boosting market growth.
- Advancements in Pediatric Drug Formulations:Innovations in drug formulations tailored for children are propelling market growth. In future, the introduction of at least 20 new pediatric-specific drugs is anticipated, focusing on improved efficacy and safety profiles. These advancements include liquid formulations and taste-masking technologies, which enhance compliance among young patients. The ongoing research and development efforts by pharmaceutical companies are crucial in addressing the unique therapeutic needs of pediatric patients, fostering a more robust market environment.
Market Challenges
- Stringent Regulatory Requirements:The regulatory landscape for pediatric drugs in Japan is notably stringent, posing challenges for manufacturers. The Pharmaceuticals and Medical Devices Agency (PMDA) mandates rigorous clinical trials specifically designed for children, which can extend the time to market. In future, the average time for pediatric drug approval is expected to be around 20 months longer than for adult drugs, creating hurdles for timely access to new treatments and increasing development costs significantly.
- Limited Availability of Pediatric Formulations:Despite the growing demand for pediatric medications, the availability of suitable formulations remains limited. In future, it is estimated that only 35% of drugs prescribed for children are specifically formulated for pediatric use. This gap often leads to off-label prescribing, which can pose safety risks. The lack of tailored formulations hampers effective treatment options, highlighting a critical challenge that needs to be addressed to improve pediatric healthcare outcomes.
Japan Pediatric Drugs Market Future Outlook
The future of the Japan pediatric drugs market appears promising, driven by ongoing advancements in drug development and increasing healthcare investments. The focus on personalized medicine is expected to gain momentum, allowing for tailored treatments that cater to individual pediatric needs. Additionally, the integration of digital health solutions will enhance patient monitoring and adherence, further supporting the growth of pediatric drug utilization. As these trends evolve, they will likely reshape the landscape of pediatric healthcare in Japan, fostering innovation and improved patient outcomes.
Market Opportunities
- Expansion of Telemedicine Services:The rise of telemedicine presents a significant opportunity for pediatric drug delivery. In future, telehealth consultations for pediatric care are projected to increase by 30%, facilitating access to healthcare services. This trend allows for timely prescriptions and follow-ups, improving medication adherence and health outcomes for children, particularly in rural areas where access to specialists is limited.
- Development of Personalized Medicine:The shift towards personalized medicine in pediatrics is gaining traction, with an expected investment of ¥12 billion ($110 million) in future for research in this area. Tailored therapies based on genetic profiles can enhance treatment efficacy and minimize adverse effects. This approach not only addresses the unique needs of pediatric patients but also opens new avenues for pharmaceutical companies to innovate and differentiate their product offerings.