Region:Global
Author(s):Dev
Product Code:KRAA6053
Pages:98
Published On:January 2026
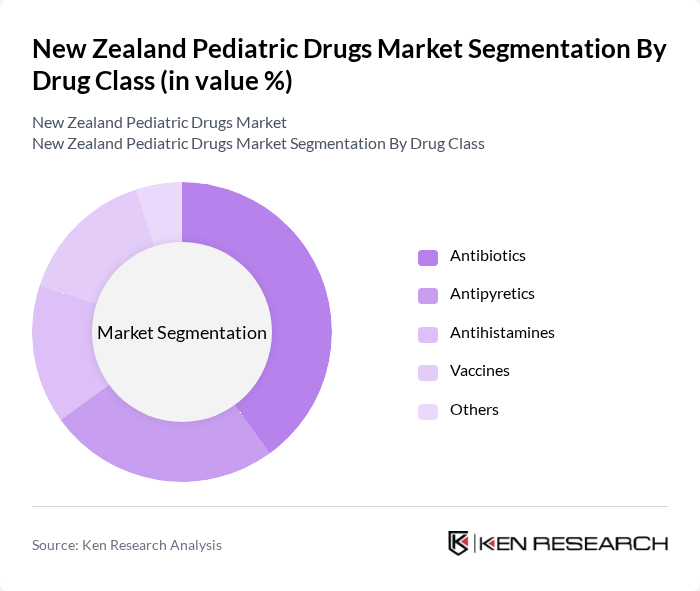
By Drug Class:The pediatric drugs market is segmented into various drug classes, including antibiotics, antipyretics, antihistamines, vaccines, and others. Among these, antibiotics are the leading sub-segment due to the high incidence of bacterial infections in children, driving demand for effective treatment options. Antipyretics also hold a significant share as they are commonly used to manage fever in pediatric patients. The increasing awareness of vaccination benefits further boosts the vaccine segment, while other drug classes cater to specific needs.
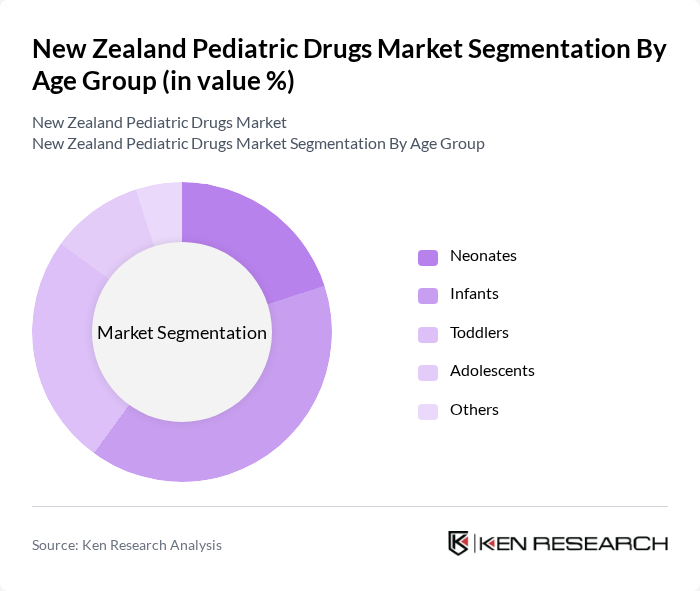
By Age Group:The market is also segmented by age groups, including neonates, infants, toddlers, adolescents, and others. Infants represent the largest segment due to their vulnerability to various health issues, necessitating a wide range of pediatric medications. Neonates also require specialized formulations, particularly for critical conditions. The toddler and adolescent segments are growing as awareness of pediatric health continues to rise, leading to increased demand for age-appropriate medications.
The New Zealand Pediatric Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Pfizer New Zealand, GlaxoSmithKline New Zealand, Novartis New Zealand, Sanofi New Zealand, Merck Sharp & Dohme New Zealand, AstraZeneca New Zealand, Johnson & Johnson New Zealand, Roche New Zealand, Teva Pharmaceuticals New Zealand, Mylan New Zealand, Amgen New Zealand, AbbVie New Zealand, Takeda Pharmaceuticals New Zealand, Bayer New Zealand, Boehringer Ingelheim New Zealand contribute to innovation, geographic expansion, and service delivery in this space.
The future of the New Zealand pediatric drugs market appears promising, driven by ongoing advancements in drug formulations and a growing emphasis on preventive healthcare. As healthcare providers increasingly adopt digital health solutions, the integration of telemedicine is expected to enhance access to pediatric care. Furthermore, the rise in pediatric clinical trials will likely foster innovation, leading to the development of more effective treatments tailored to children's unique health needs, ultimately improving health outcomes across the population.
| Segment | Sub-Segments |
|---|---|
| By Drug Class | Antibiotics Antipyretics Antihistamines Vaccines Others |
| By Age Group | Neonates Infants Toddlers Adolescents Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Direct Sales Others |
| By Formulation Type | Liquid Tablet Injection Topical Others |
| By Therapeutic Area | Infectious Diseases Respiratory Disorders Neurological Disorders Gastrointestinal Disorders Others |
| By Region | North Island South Island Others |
| By Market Type | Prescription Drugs Over-the-Counter Drugs Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pediatricians and Child Health Specialists | 45 | Pediatricians, Family Doctors |
| Pharmacists in Community Settings | 80 | Community Pharmacists, Pharmacy Managers |
| Parents of Pediatric Patients | 120 | Parents, Guardians |
| Healthcare Policy Makers | 50 | Health Administrators, Policy Analysts |
| Hospital Administrators | 70 | Hospital Managers, Clinical Directors |
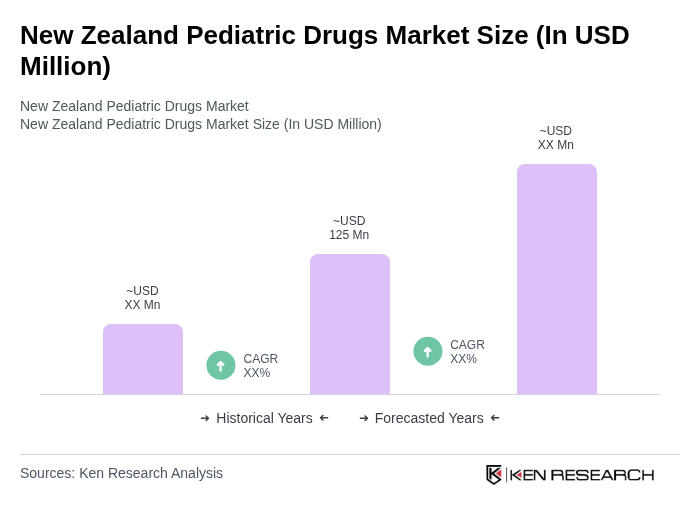
The New Zealand Pediatric Drugs Market is valued at approximately USD 125 million, reflecting a significant growth driven by increasing healthcare expenditure, rising prevalence of pediatric diseases, and advancements in drug formulations specifically designed for children.