Region:Asia
Author(s):Geetanshi
Product Code:KRAE1973
Pages:83
Published On:February 2026
By Type:
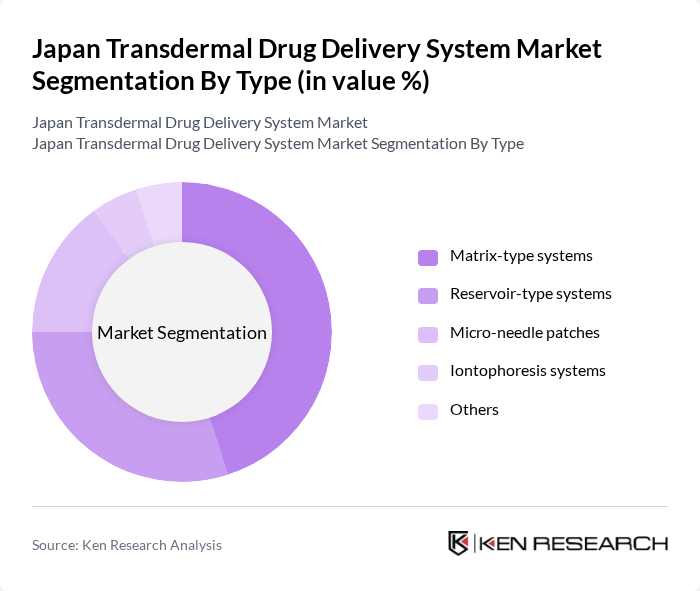
The market is segmented into various types, including matrix-type systems, reservoir-type systems, micro-needle patches, iontophoresis systems, and others. Among these, matrix-type systems are currently leading the market due to their ease of use and effectiveness in delivering a wide range of drugs. The growing preference for self-administration and the increasing number of approved matrix-type products are driving their dominance. Reservoir-type systems also hold a significant share, particularly for drugs requiring controlled release profiles.
By End-User:
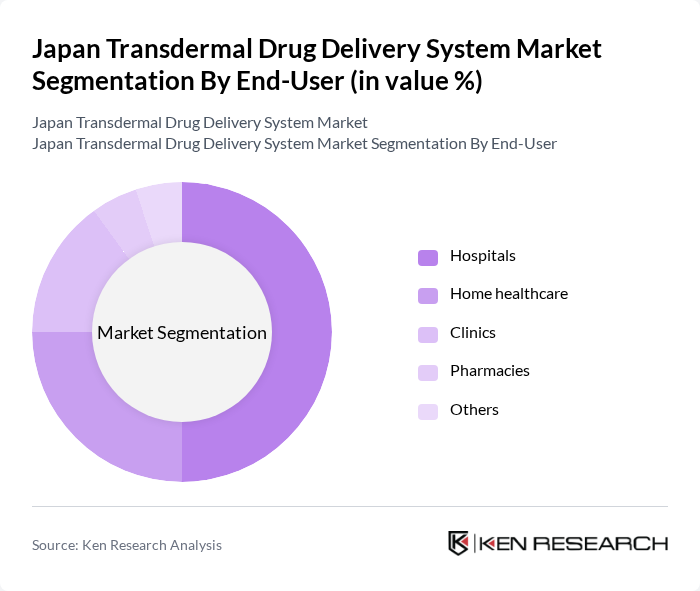
The end-user segmentation includes hospitals, home healthcare, clinics, pharmacies, and others. Hospitals are the leading end-user segment, driven by the increasing adoption of transdermal systems for pain management and chronic disease treatment. Home healthcare is also gaining traction as patients prefer convenient drug delivery methods that can be administered at home. The rise in outpatient care and the growing trend of self-medication are further propelling the demand in this segment.
The Japan Transdermal Drug Delivery System Market is characterized by a dynamic mix of regional and international players. Leading participants such as Hisamitsu Pharmaceutical Co., Inc., Teijin Pharma Limited, UCB Japan Co., Ltd., Novartis Pharma K.K., Johnson & Johnson K.K., Pfizer Japan Inc., Daiichi Sankyo Company, Limited, Astellas Pharma Inc., Takeda Pharmaceutical Company Limited, Sato Pharmaceutical Co., Ltd., Otsuka Pharmaceutical Co., Ltd., Mitsubishi Tanabe Pharma Corporation, Kyowa Kirin Co., Ltd., Eisai Co., Ltd., Sumitomo Dainippon Pharma Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the transdermal drug delivery system market in Japan appears promising, driven by ongoing innovations and a growing focus on patient-centric solutions. As healthcare providers increasingly adopt digital health technologies, the integration of these advancements into transdermal systems is expected to enhance patient engagement and adherence. Furthermore, the rising demand for personalized medicine will likely spur the development of tailored transdermal therapies, creating new avenues for growth and improving patient outcomes in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Matrix-type systems Reservoir-type systems Micro-needle patches Iontophoresis systems Others |
| By End-User | Hospitals Home healthcare Clinics Pharmacies Others |
| By Application | Pain management Hormonal therapies Cardiovascular treatments Neurological disorders Others |
| By Drug Type | Prescription drugs Over-the-counter drugs Biologics Others |
| By Distribution Channel | Online pharmacies Retail pharmacies Hospital pharmacies Others |
| By Region | Kanto Kansai Chubu Kyushu Others |
| By Technology | Passive transdermal systems Active transdermal systems Hybrid systems Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 100 | Product Managers, R&D Directors |
| Healthcare Providers | 80 | Dermatologists, Pharmacists |
| Patients Using Transdermal Systems | 120 | Chronic Pain Patients, Diabetes Patients |
| Regulatory Bodies | 50 | Regulatory Affairs Specialists, Compliance Officers |
| Market Analysts | 70 | Healthcare Market Analysts, Industry Consultants |
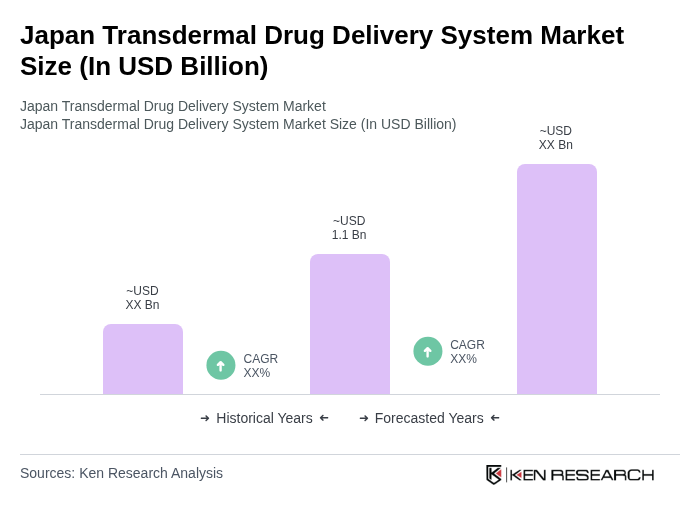
The Japan Transdermal Drug Delivery System market is valued at approximately USD 1.1 billion, reflecting a significant growth trend driven by the increasing prevalence of chronic diseases and advancements in drug delivery technologies.