Region:Global
Author(s):Geetanshi
Product Code:KRAE1974
Pages:81
Published On:February 2026
By Technology:The technology segment includes various methods used in transdermal drug delivery systems. The subsegments are Iontophoresis, Electroporation, Radio Frequency, Ultrasound, Microneedle Arrays, and Others. Each of these technologies offers unique advantages in terms of drug delivery efficiency and patient comfort.
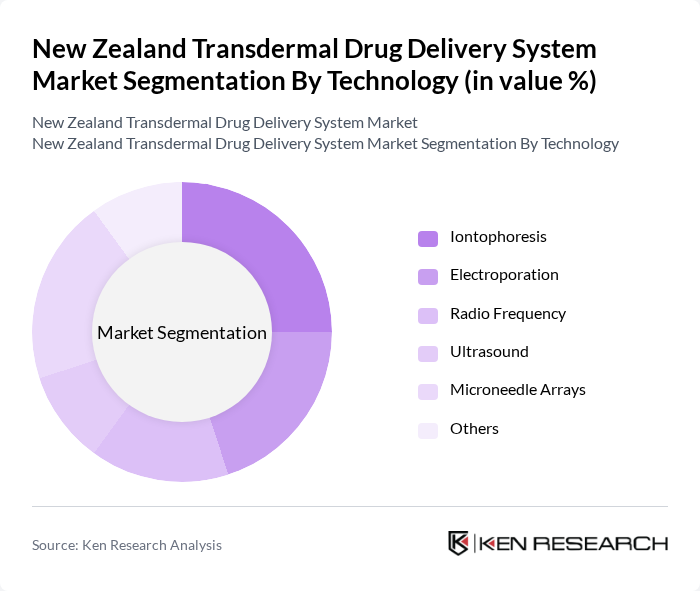
The Iontophoresis subsegment is currently dominating the market due to its ability to enhance drug delivery through the skin using electrical currents. This method is particularly effective for delivering macromolecules and has gained popularity among patients for its non-invasive nature. The increasing focus on patient-centric healthcare solutions and the growing prevalence of chronic conditions are further driving the adoption of this technology.
By Application:The application segment encompasses various therapeutic areas where transdermal drug delivery systems are utilized. The subsegments include Pain Management, Cardiovascular Diseases, Central Nervous System Disorders, Hormonal Therapies, Gastrointestinal Conditions, Cancer Therapies, and Others. Each application area reflects the diverse uses of transdermal systems in modern medicine.

The Pain Management application is leading the market, driven by the increasing prevalence of chronic pain conditions and the growing preference for non-invasive treatment options. Transdermal systems provide effective pain relief while minimizing side effects associated with oral medications, making them a popular choice among patients and healthcare providers alike.
The New Zealand Transdermal Drug Delivery System Market is characterized by a dynamic mix of regional and international players. Leading participants such as Hisamitsu Pharmaceutical, Pfizer New Zealand, Novartis New Zealand, Johnson & Johnson New Zealand, Teva Pharmaceuticals New Zealand, Sandoz New Zealand, GlaxoSmithKline New Zealand, Merck Sharp & Dohme New Zealand, Sanofi New Zealand, AFT Pharmaceuticals, Douglas Pharmaceuticals, Pacific Edge contribute to innovation, geographic expansion, and service delivery in this space.
The future of the transdermal drug delivery system market in New Zealand appears promising, driven by technological advancements and a growing focus on patient-centric solutions. As healthcare providers increasingly adopt digital health technologies, the integration of these innovations into transdermal systems is expected to enhance patient monitoring and adherence. Additionally, the rising interest in personalized medicine will likely lead to tailored transdermal therapies, addressing specific patient needs and improving treatment outcomes in future.
| Segment | Sub-Segments |
|---|---|
| By Technology | Iontophoresis Electroporation Radio Frequency Ultrasound Microneedle Arrays Others |
| By Application | Pain Management Cardiovascular Diseases Central Nervous System Disorders Hormonal Therapies Gastrointestinal Conditions Cancer Therapies Others |
| By Product Type | Transdermal Patches Transdermal Gels Transdermal Sprays Others |
| By End-User | Hospitals Retail Pharmacies Online Pharmacies Home Healthcare Clinics Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Others |
| By Region | North Island South Island |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Professionals | 120 | Pharmacists, General Practitioners, Dermatologists |
| Patients Using Transdermal Systems | 100 | Chronic Pain Patients, Hormone Therapy Patients |
| Pharmaceutical Manufacturers | 80 | R&D Managers, Product Development Leads |
| Regulatory Experts | 50 | Compliance Officers, Regulatory Affairs Specialists |
| Market Analysts | 60 | Healthcare Market Researchers, Industry Analysts |
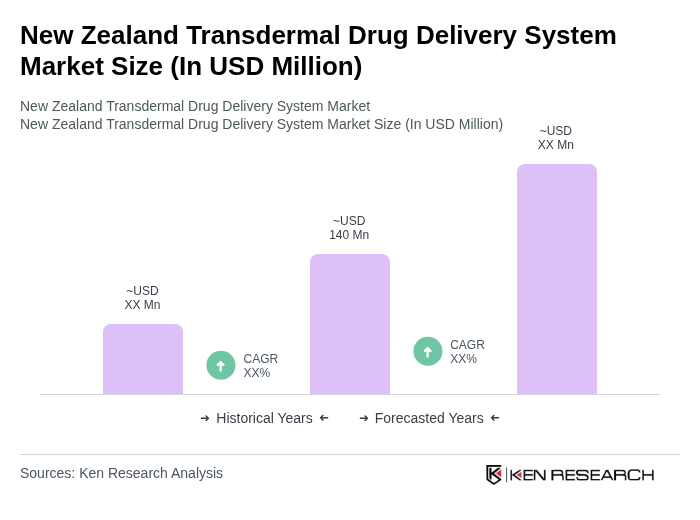
The New Zealand Transdermal Drug Delivery System market is valued at approximately USD 140 million, reflecting a five-year historical analysis. This growth is driven by the rising prevalence of chronic diseases and advancements in transdermal technology.