Region:Middle East
Author(s):Dev
Product Code:KRAA3771
Pages:95
Published On:January 2026
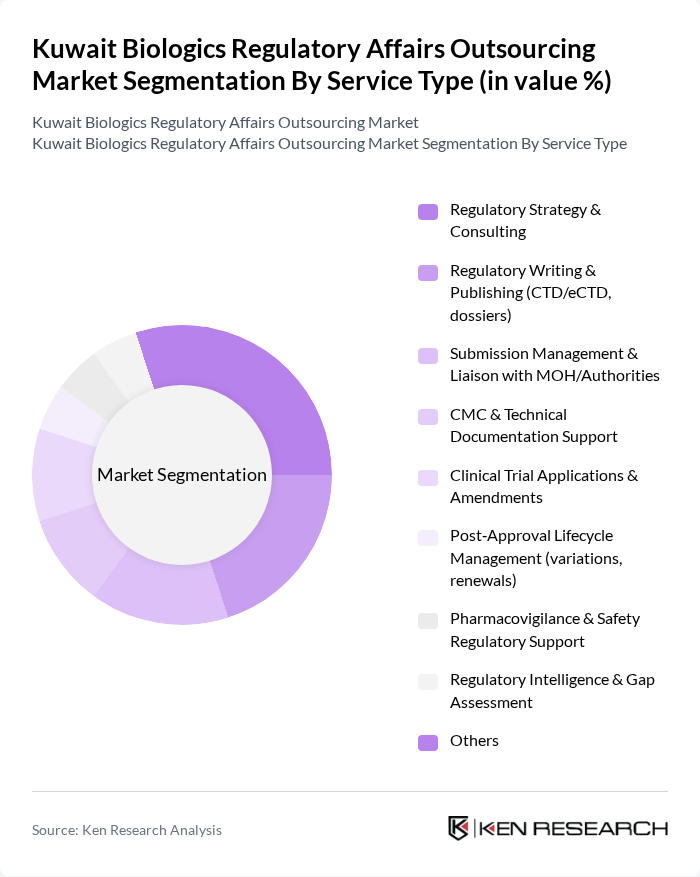
By Service Type:The service type segmentation includes various essential services that support the regulatory process for biologics. Key services include Regulatory Strategy & Consulting, which helps companies navigate the complex, evolving regulatory landscape and align development programs with Kuwait Ministry of Health and GCC requirements; Regulatory Writing & Publishing, which ensures that all documentation, including CTD/eCTD dossiers and technical reports, meets international and local regulatory standards; and Submission Management & Liaison with MOH/Authorities, which facilitates end?to?end coordination of submissions, responses to queries, and interactions with regulators. Other services such as CMC & Technical Documentation Support, Clinical Trial Applications & Amendments (including support for multinational biologics trials entering Kuwait), and Pharmacovigilance & Safety Regulatory Support are also critical in ensuring compliance and safety throughout the product lifecycle, especially as biologics and biosimilars portfolios expand and post?marketing safety expectations increase.
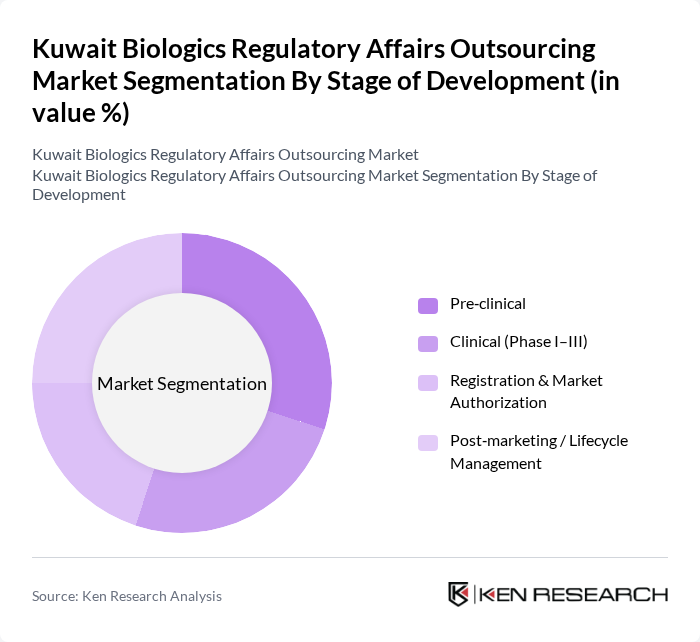
By Stage of Development:The stage of development segmentation encompasses the various phases that biologics undergo from inception to market entry. This includes Pre-clinical, where initial research, in?vitro and in?vivo studies, and early CMC development occur; Clinical (Phase I–III), which involves testing the biologic in human subjects and requires protocol approvals, import licenses, and ongoing safety submissions; Registration & Market Authorization, where companies seek approval from regulatory bodies through full CTD/eCTD dossiers, pricing, and labeling negotiations; and Post-marketing / Lifecycle Management, which ensures ongoing compliance, safety reporting, periodic safety update reports, variations, and renewals after the product has been launched. Each stage is crucial for ensuring that biologics are safe and effective for public use and increasingly relies on specialized outsourcing partners to manage documentation, submissions, and interactions with authorities efficiently.
The Kuwait Biologics Regulatory Affairs Outsourcing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Kuwait Life Sciences Company (KLSC), Kuwait Pharmaceutical Industries Company (KPI), Kuwait Saudi Pharmaceutical Industries Company (KSPICO), Kuwait University – Health Sciences Center & Clinical Research Unit, Ministry of Health, Kuwait – Drug & Food Control / Registration Department, Kuwait Institute for Scientific Research (KISR), Local Specialized Regulatory Affairs & Pharmacovigilance Consultancies, Regional CROs & Biologics RA Outsourcing Firms Active in Kuwait, GCC?based Biotech & Biopharma Sponsors Active in Kuwait, Multinational CROs / CDMOs with Kuwait Biologics RA Footprint, Kuwait Cancer Control Center (KCCC), Major Private Hospital Groups in Kuwait (e.g., Al?Sabah, Royale Hayat, Dar Al Shifa), Kuwait University Hospital & Teaching Hospitals Network, Kuwait Foundation for the Advancement of Sciences (KFAS) – Health & Biotech Programs, Other Key Institutional Stakeholders & Industry Associations contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Kuwait biologics regulatory affairs outsourcing market appears promising, driven by increasing healthcare investments and a growing focus on personalized medicine. As the demand for innovative therapies rises, companies will likely seek to streamline their regulatory processes through outsourcing. Additionally, advancements in digital solutions and artificial intelligence will enhance compliance and efficiency, enabling firms to navigate the complex regulatory landscape more effectively while ensuring patient safety and product efficacy.
| Segment | Sub-Segments |
|---|---|
| By Service Type | Regulatory Strategy & Consulting Regulatory Writing & Publishing (CTD/eCTD, dossiers) Submission Management & Liaison with MOH/Authorities CMC & Technical Documentation Support Clinical Trial Applications & Amendments Post?Approval Lifecycle Management (variations, renewals) Pharmacovigilance & Safety Regulatory Support Regulatory Intelligence & Gap Assessment Others |
| By Stage of Development | Pre?clinical Clinical (Phase I–III) Registration & Market Authorization Post?marketing / Lifecycle Management |
| By Biologic Type | Monoclonal Antibodies Vaccines Recombinant Proteins & Cytokines Cell & Gene Therapies Biosimilars Others |
| By End-User | Multinational Pharmaceutical Companies Local & Regional Pharmaceutical Manufacturers Biotechnology Firms Contract Research Organizations (CROs) & CDMOs Medical Device & Diagnostics Companies (biologic?related) Government & Academic / Research Institutions Others |
| By Service Model / Engagement Model | Full?Service Outsourcing Functional Service Provider (FSP) Project?Based Engagements Dedicated / Long?term Retainer Teams Hybrid Models Others |
| By Client Size | Large Enterprises Medium Enterprises Small & Emerging Biotech Others |
| By Region | Kuwait City & Capital Governorate Hawalli & Farwaniya Ahmadi Jahra Mubarak Al?Kabeer |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biologics Regulatory Compliance | 60 | Regulatory Affairs Managers, Compliance Officers |
| Clinical Trials Management | 50 | Clinical Research Coordinators, Project Managers |
| Market Access Strategies | 40 | Market Access Managers, Health Economists |
| Pharmaceutical Quality Assurance | 40 | Quality Control Managers, Production Supervisors |
| Healthcare Policy Impact | 50 | Healthcare Policy Analysts, Biopharmaceutical Executives |
The Kuwait Biologics Regulatory Affairs Outsourcing Market is valued at approximately USD 7 million, reflecting a historical analysis and alignment with country-level biologics regulatory affairs data.