Malaysia Nucleic Acid Based Therapeutics Market Overview
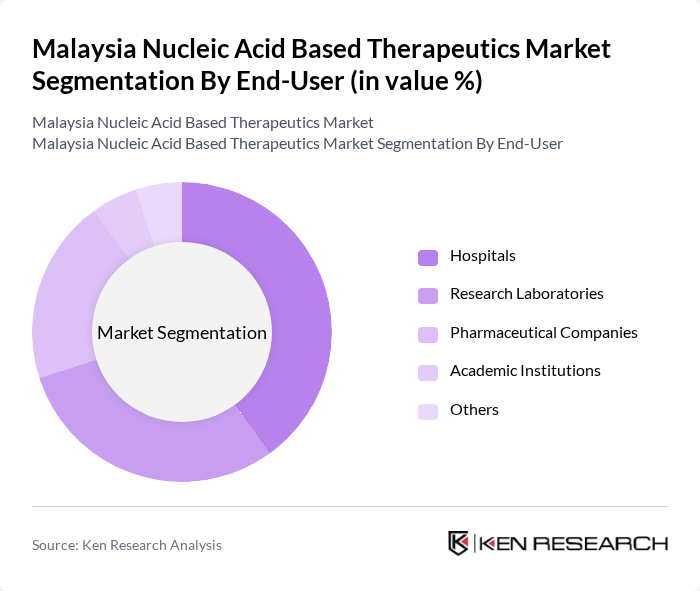
- The Malaysia Nucleic Acid Based Therapeutics Market is valued at USD 1.2 billion, based on a five-year historical analysis. This growth is primarily driven by advancements in genetic research, increasing prevalence of genetic disorders, and rising investments in biotechnology. The market is also supported by a growing demand for personalized medicine and innovative treatment options that leverage nucleic acid technologies.
- Key players in this market include Kuala Lumpur, Penang, and Johor Bahru. These cities dominate due to their robust healthcare infrastructure, presence of research institutions, and supportive government policies that foster innovation in biotechnology and pharmaceuticals. The concentration of skilled professionals and access to funding further enhance their competitive edge in the nucleic acid therapeutics sector.
- In 2023, the Malaysian government implemented the National Biotechnology Policy, which aims to promote the development and commercialization of biotechnology products, including nucleic acid-based therapeutics. This policy includes incentives for research and development, regulatory support, and funding opportunities to encourage local and foreign investments in the biotechnology sector.
Malaysia Nucleic Acid Based Therapeutics Market Segmentation
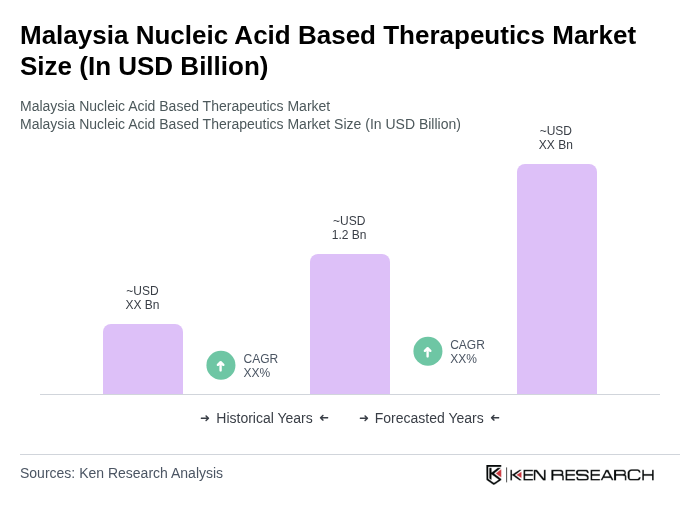
By Type:The market is segmented into various types, including Antisense Oligonucleotides, RNA Interference (RNAi), CRISPR/Cas9, DNA Vaccines, and Others. Among these, Antisense Oligonucleotides are gaining traction due to their targeted therapeutic applications in treating genetic disorders and cancers. RNAi technologies are also witnessing significant adoption for their potential in gene silencing and therapeutic interventions. The CRISPR/Cas9 segment is emerging as a revolutionary tool for gene editing, while DNA vaccines are being explored for their efficacy in infectious disease prevention.
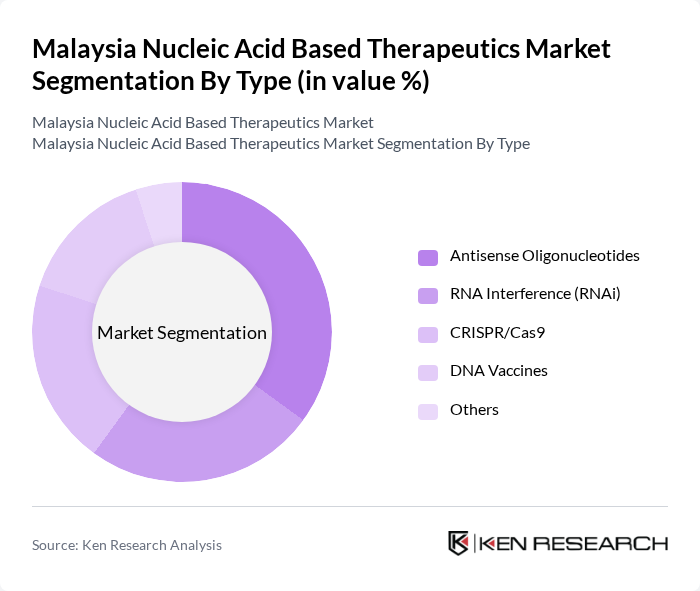
By End-User:The end-user segmentation includes Hospitals, Research Laboratories, Pharmaceutical Companies, Academic Institutions, and Others. Hospitals are the leading end-users due to the increasing adoption of nucleic acid-based therapies for patient treatment. Research laboratories are also significant contributors, focusing on the development and testing of new therapeutics. Pharmaceutical companies are investing heavily in R&D to bring innovative products to market, while academic institutions play a crucial role in foundational research and training the next generation of scientists.
Malaysia Nucleic Acid Based Therapeutics Market Competitive Landscape
The Malaysia Nucleic Acid Based Therapeutics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Biocon Limited, Genentech, Inc., Moderna, Inc., Novartis AG, GSK (GlaxoSmithKline), Pfizer Inc., Roche Holding AG, Amgen Inc., Regeneron Pharmaceuticals, Inc., Sangamo Therapeutics, Inc., Vertex Pharmaceuticals Incorporated, Alnylam Pharmaceuticals, Inc., Ionis Pharmaceuticals, Inc., Sarepta Therapeutics, Inc., CRISPR Therapeutics AG contribute to innovation, geographic expansion, and service delivery in this space.
Malaysia Nucleic Acid Based Therapeutics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Genetic Disorders:The rise in genetic disorders in Malaysia is a significant growth driver for nucleic acid-based therapeutics. According to the Ministry of Health Malaysia, approximately 1 in 500 individuals is affected by a genetic disorder, translating to around 62,000 cases annually. This increasing prevalence necessitates innovative treatment options, propelling demand for nucleic acid therapies. Furthermore, the growing awareness of genetic testing and counseling is expected to enhance early diagnosis, further driving market growth.
- Advancements in Gene Editing Technologies:The rapid advancements in gene editing technologies, such as CRISPR-Cas9, are transforming the landscape of nucleic acid-based therapeutics in Malaysia. The Malaysian Biotechnology Corporation reported that investments in gene editing research reached RM 550 million in future. These technologies enable precise modifications to genetic material, enhancing the efficacy of treatments for various genetic disorders. As these technologies become more accessible, they are expected to significantly boost the development of innovative therapies in the market.
- Rising Investment in Biotechnology Research:Malaysia's biotechnology sector is witnessing increased investment, which is a crucial driver for the nucleic acid-based therapeutics market. In future, the government allocated RM 1.3 billion to support biotechnology research and development initiatives. This funding is aimed at fostering innovation and facilitating the commercialization of new therapies. The influx of capital is expected to accelerate research activities, leading to the development of novel nucleic acid-based treatments that address unmet medical needs.
Market Challenges
- High Cost of Development and Production:One of the primary challenges facing the nucleic acid-based therapeutics market in Malaysia is the high cost associated with development and production. The average cost to develop a new therapeutic can exceed RM 1.2 billion, which poses a significant barrier for many companies. This financial burden can limit the number of new entrants in the market and slow down the pace of innovation, ultimately affecting patient access to these advanced therapies.
- Regulatory Hurdles and Lengthy Approval Processes:The regulatory landscape for nucleic acid-based therapeutics in Malaysia presents significant challenges. The approval process for new therapies can take several years, with the Malaysian National Pharmaceutical Regulatory Agency (NPRA) requiring extensive clinical trial data. In future, the average time for drug approval was reported at 26 months, which can delay market entry for innovative therapies. These regulatory hurdles can deter investment and slow the overall growth of the market.
Malaysia Nucleic Acid Based Therapeutics Market Future Outlook
The future of the nucleic acid-based therapeutics market in Malaysia appears promising, driven by technological advancements and increasing healthcare investments. The integration of artificial intelligence in drug discovery is expected to streamline the development process, enhancing efficiency and reducing costs. Additionally, the growing focus on personalized medicine will likely lead to tailored therapies that meet specific patient needs. As the market evolves, collaboration between biotech firms and academic institutions will further accelerate innovation and improve patient outcomes.
Market Opportunities
- Expansion of Personalized Medicine:The shift towards personalized medicine presents a significant opportunity for the nucleic acid-based therapeutics market. With the increasing demand for tailored treatments, companies can leverage genetic insights to develop therapies that cater to individual patient profiles. This approach not only enhances treatment efficacy but also improves patient adherence, potentially leading to better health outcomes and increased market share for innovative therapies.
- Collaborations with Academic Institutions:Collaborations between biotech companies and academic institutions are poised to drive innovation in the nucleic acid-based therapeutics market. These partnerships can facilitate access to cutting-edge research, resources, and expertise, accelerating the development of new therapies. In future, several Malaysian universities reported joint research initiatives with biotech firms, highlighting the potential for groundbreaking discoveries that can enhance the therapeutic landscape.