Region:Asia
Author(s):Rebecca
Product Code:KRAE2515
Pages:93
Published On:February 2026
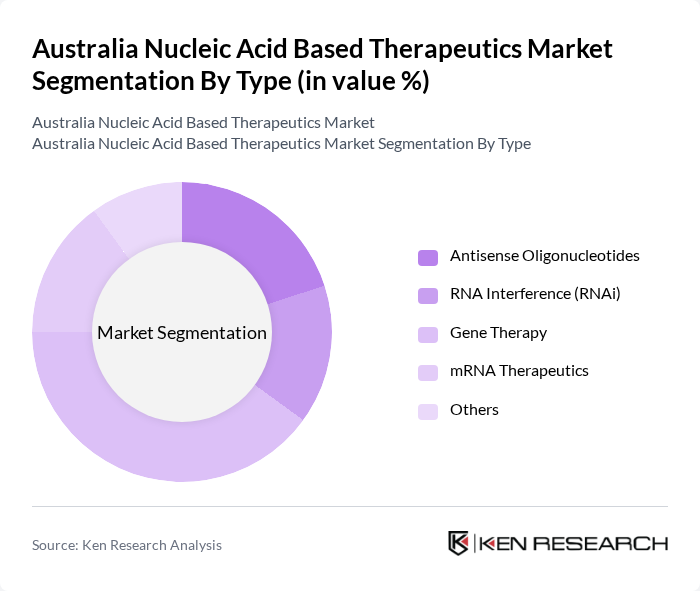
By Type:The market is segmented into various types, including Antisense Oligonucleotides, RNA Interference (RNAi), Gene Therapy, mRNA Therapeutics, and Others. Among these, Gene Therapy is currently the leading sub-segment due to its transformative potential in treating genetic disorders and its increasing application in oncology. The growing number of successful clinical trials and approvals for gene therapies has significantly boosted its market presence.
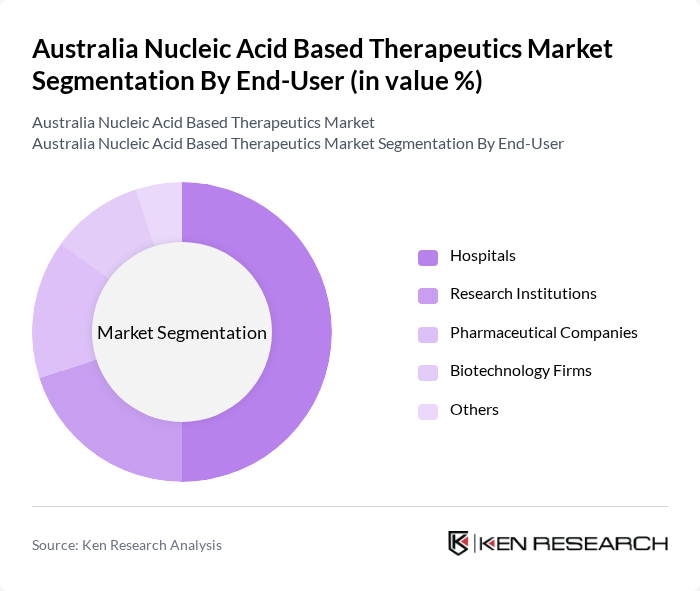
By End-User:The end-user segmentation includes Hospitals, Research Institutions, Pharmaceutical Companies, Biotechnology Firms, and Others. Hospitals are the dominant end-user segment, driven by the increasing adoption of advanced therapies and the need for specialized treatment options. The growing number of patients seeking innovative therapies in hospital settings has led to a significant rise in demand for nucleic acid-based therapeutics.
The Australia Nucleic Acid Based Therapeutics Market is characterized by a dynamic mix of regional and international players. Leading participants such as CSL Limited, Novogen Limited, Genetic Technologies Limited, Sirtex Medical Limited, Antisense Therapeutics Limited, Imugene Limited, Opthea Limited, Mesoblast Limited, Paradigm Biopharmaceuticals Limited, Avita Medical Limited, Clinuvel Pharmaceuticals Limited, Progen Pharmaceuticals Limited, Neuren Pharmaceuticals Limited, Cyclopharm Limited, AFT Pharmaceuticals Limited contribute to innovation, geographic expansion, and service delivery in this space.
The future of the nucleic acid-based therapeutics market in Australia appears promising, driven by ongoing advancements in gene editing technologies and increased public-private partnerships. As the healthcare landscape shifts towards personalized medicine, the demand for tailored therapies is expected to rise. Furthermore, the integration of artificial intelligence in drug discovery processes is likely to enhance efficiency and innovation, paving the way for novel therapeutic solutions that address unmet medical needs.
| Segment | Sub-Segments |
|---|---|
| By Type | Antisense Oligonucleotides RNA Interference (RNAi) Gene Therapy mRNA Therapeutics Others |
| By End-User | Hospitals Research Institutions Pharmaceutical Companies Biotechnology Firms Others |
| By Application | Oncology Infectious Diseases Genetic Disorders Cardiovascular Diseases Others |
| By Delivery Method | Viral Vectors Non-Viral Vectors Lipid Nanoparticles Others |
| By Region | New South Wales Victoria Queensland Western Australia Others |
| By Research Phase | Preclinical Clinical Trials Marketed Products Others |
| By Funding Source | Government Grants Private Investments Venture Capital Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Therapeutics | 100 | Oncologists, Clinical Researchers |
| Genetic Disorder Treatments | 80 | Geneticists, Rare Disease Specialists |
| Infectious Disease Applications | 70 | Infectious Disease Physicians, Public Health Officials |
| Regulatory Insights | 60 | Regulatory Affairs Managers, Compliance Officers |
| Patient Advocacy Perspectives | 50 | Patient Advocates, Healthcare Policy Makers |
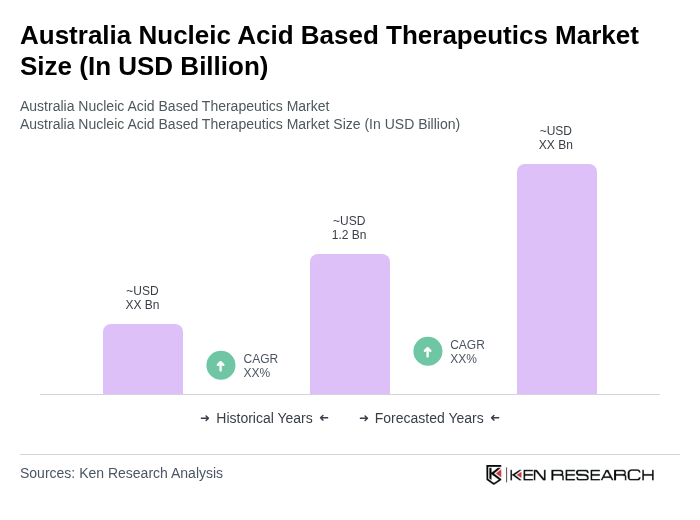
The Australia Nucleic Acid Based Therapeutics Market is valued at approximately USD 1.2 billion, reflecting significant growth driven by advancements in genetic research, increasing prevalence of genetic disorders, and rising investments in biotechnology.