Oman Nucleic Acid Based Therapeutics Market Overview
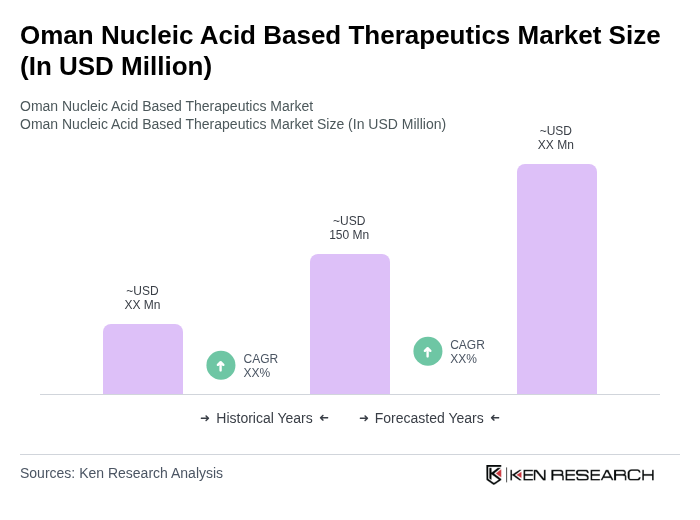
- The Oman Nucleic Acid Based Therapeutics Market is valued at USD 150 million, based on a five-year historical analysis. This growth is primarily driven by advancements in genetic research, increasing prevalence of genetic disorders, and rising investments in biotechnology. The market is also supported by a growing awareness of personalized medicine and the potential of nucleic acid-based therapies to address unmet medical needs.
- Key cities such as Muscat and Salalah dominate the market due to their well-established healthcare infrastructure and research facilities. Muscat, being the capital, attracts significant investments and talent, while Salalah benefits from its strategic location and access to international markets, fostering collaboration between local and global biotech firms.
- In 2023, the Omani government implemented a regulatory framework aimed at promoting the development and commercialization of nucleic acid-based therapeutics. This framework includes guidelines for clinical trials, manufacturing standards, and market authorization processes, ensuring that therapies meet safety and efficacy requirements while encouraging innovation in the biotechnology sector.
Oman Nucleic Acid Based Therapeutics Market Segmentation
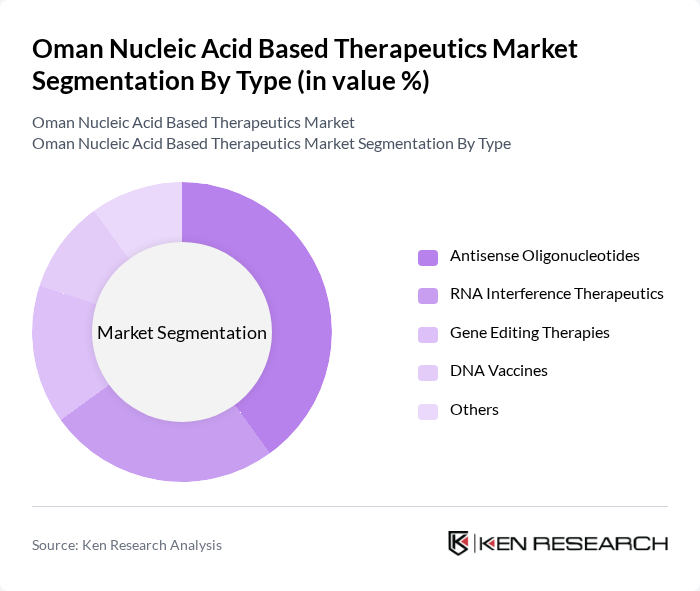
By Type:The market is segmented into various types of nucleic acid-based therapeutics, including Antisense Oligonucleotides, RNA Interference Therapeutics, Gene Editing Therapies, DNA Vaccines, and Others. Among these, Antisense Oligonucleotides are currently leading the market due to their effectiveness in treating genetic disorders and their growing application in oncology. The increasing number of clinical trials and approvals for antisense therapies is driving their dominance.
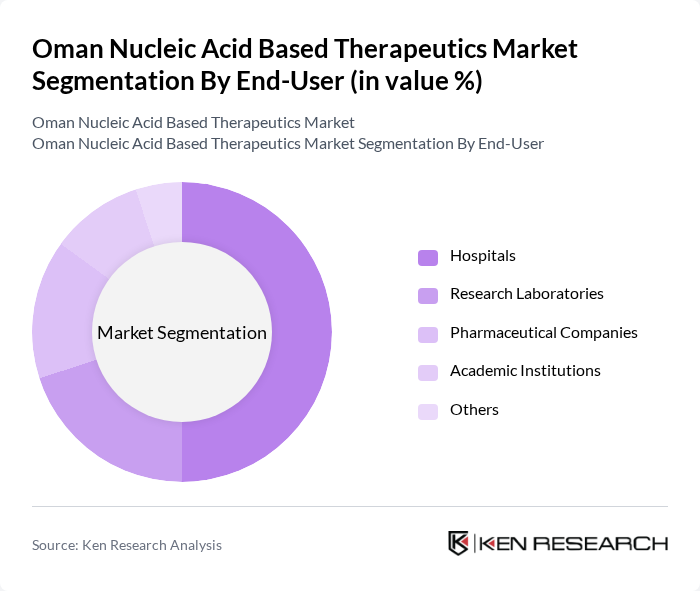
By End-User:The end-user segmentation includes Hospitals, Research Laboratories, Pharmaceutical Companies, Academic Institutions, and Others. Hospitals are the leading end-user segment, driven by the increasing adoption of advanced therapies and the need for specialized treatment options. The growing number of patients seeking innovative therapies in hospitals is significantly contributing to this segment's growth.
Oman Nucleic Acid Based Therapeutics Market Competitive Landscape
The Oman Nucleic Acid Based Therapeutics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis AG, Gilead Sciences, Inc., Amgen Inc., Regeneron Pharmaceuticals, Inc., Vertex Pharmaceuticals Incorporated, BioMarin Pharmaceutical Inc., Sarepta Therapeutics, Inc., Moderna, Inc., Illumina, Inc., CRISPR Therapeutics AG, Editas Medicine, Inc., Alnylam Pharmaceuticals, Inc., Sangamo Therapeutics, Inc., Cellectis S.A., Intellia Therapeutics, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
Oman Nucleic Acid Based Therapeutics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Genetic Disorders:The prevalence of genetic disorders in Oman is rising, with approximately 1 in 1,000 births affected by conditions such as cystic fibrosis and muscular dystrophy. According to the Oman Ministry of Health, around 2,200 new cases of genetic disorders are reported annually. This growing incidence drives demand for nucleic acid-based therapeutics, as healthcare providers seek innovative solutions to address these complex health challenges effectively.
- Advancements in Gene Editing Technologies:The global gene editing market is projected to reach $12 billion in the future, with CRISPR technology leading the way. In Oman, local research institutions are increasingly adopting these technologies, evidenced by a 35% increase in gene editing research projects over the past two years. This trend enhances the development of nucleic acid-based therapies, enabling more precise and effective treatments for genetic disorders and other diseases.
- Rising Investment in Biotechnology Research:Oman’s government has allocated approximately $250 million for biotechnology research in the future, reflecting a commitment to advancing healthcare solutions. This funding supports the development of nucleic acid-based therapeutics, fostering innovation and collaboration among local universities and biotech firms. As a result, the market is expected to benefit from enhanced research capabilities and the introduction of novel therapies tailored to the Omani population's needs.
Market Challenges
- High Cost of Development and Production:The average cost of developing a new nucleic acid-based therapy can exceed $1.2 billion, which poses a significant barrier for companies operating in Oman. This high financial burden limits the number of firms willing to invest in research and development, ultimately slowing the introduction of innovative therapies to the market. As a result, many potential treatments remain underdeveloped or delayed.
- Regulatory Hurdles and Compliance Issues:The regulatory landscape for nucleic acid-based therapeutics in Oman is complex, with stringent guidelines that can delay product approvals. The Oman Food and Drug Authority (OFDA) has reported an average approval time of 20 months for new therapies. These regulatory challenges can deter investment and slow the pace of innovation, hindering the growth of the nucleic acid-based therapeutics market in the region.
Oman Nucleic Acid Based Therapeutics Market Future Outlook
The future of the nucleic acid-based therapeutics market in Oman appears promising, driven by technological advancements and increased healthcare investments. As gene editing technologies continue to evolve, the potential for innovative therapies will expand. Additionally, the growing focus on personalized medicine will likely lead to tailored treatment options for patients, enhancing therapeutic efficacy. Collaborative efforts between government, academia, and industry will further accelerate research and development, positioning Oman as a key player in the biotechnology sector.
Market Opportunities
- Expansion of Healthcare Infrastructure:Oman is investing $2 billion in healthcare infrastructure improvements in the future. This expansion will enhance access to advanced medical treatments, including nucleic acid-based therapies, thereby increasing patient uptake and driving market growth. Improved facilities will also facilitate clinical trials, fostering innovation in the sector.
- Collaborations with Research Institutions:Partnerships between biotech firms and local universities are on the rise, with over 20 active collaborations reported in the future. These alliances are crucial for knowledge exchange and resource sharing, enabling the development of cutting-edge nucleic acid-based therapies. Such collaborations can significantly enhance research capabilities and accelerate the commercialization of new treatments.