Region:Asia
Author(s):Rebecca
Product Code:KRAE2509
Pages:94
Published On:February 2026
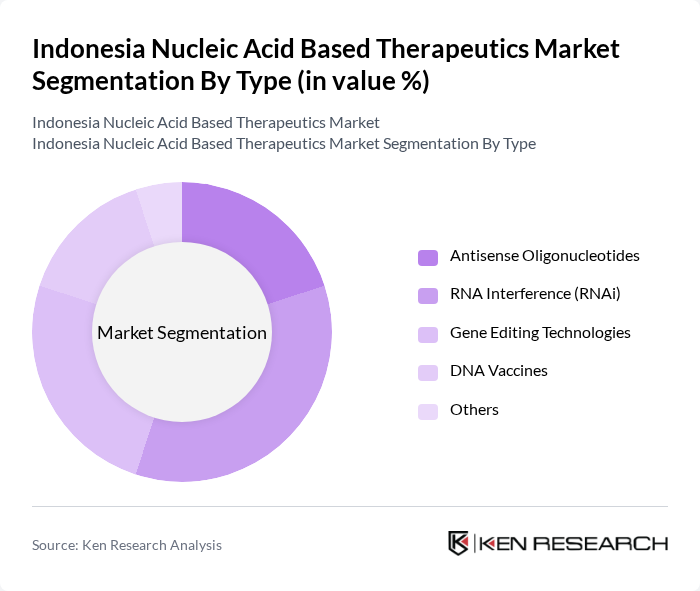
By Type:The market is segmented into various types of nucleic acid-based therapeutics, including Antisense Oligonucleotides, RNA Interference (RNAi), Gene Editing Technologies, DNA Vaccines, and Others. Among these, RNA Interference (RNAi) is currently the leading sub-segment due to its effectiveness in silencing specific genes associated with diseases, making it a preferred choice for therapeutic applications. The growing focus on precision medicine and targeted therapies further drives the demand for RNAi technologies.
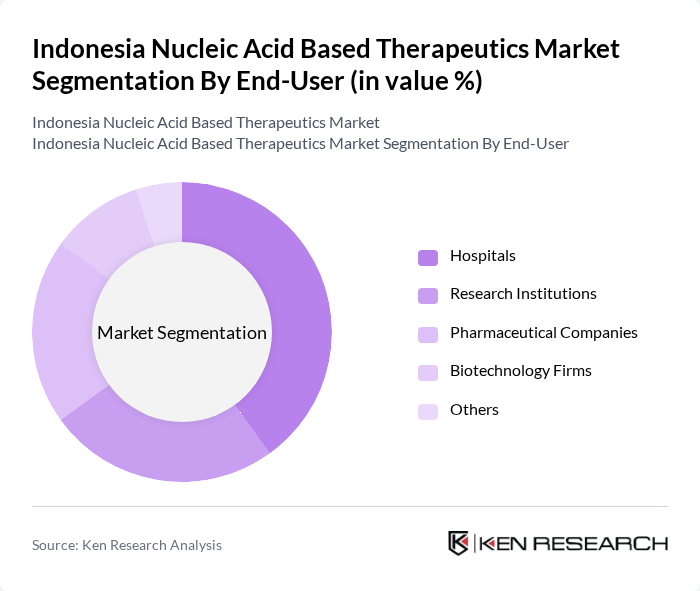
By End-User:The end-user segmentation includes Hospitals, Research Institutions, Pharmaceutical Companies, Biotechnology Firms, and Others. Hospitals are the dominant end-user segment, driven by the increasing adoption of advanced therapeutic options for treating complex diseases. The rising number of clinical trials and collaborations between hospitals and biotech firms further enhance the demand for nucleic acid-based therapeutics in clinical settings.
The Indonesia Nucleic Acid Based Therapeutics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Kalbe Farma, Bio Farma, Indofarma, Kimia Farma, PT. Medisafe Technologies, PT. Genetika Science, PT. BioNexus, PT. Eijkman Institute for Molecular Biology, PT. Siloam Hospitals, PT. Novartis Indonesia, PT. Roche Indonesia, PT. Merck Sharp & Dohme Indonesia, PT. Pfizer Indonesia, PT. Sanofi Indonesia, PT. AstraZeneca Indonesia contribute to innovation, geographic expansion, and service delivery in this space.
The future of the nucleic acid-based therapeutics market in Indonesia appears promising, driven by advancements in technology and increasing healthcare demands. As the prevalence of genetic disorders rises, the focus on personalized medicine is expected to intensify. Additionally, the integration of artificial intelligence in drug development processes will likely enhance efficiency and reduce time-to-market for new therapies. These trends indicate a robust growth trajectory for the sector, fostering innovation and improving patient outcomes in the future.
| Segment | Sub-Segments |
|---|---|
| By Type | Antisense Oligonucleotides RNA Interference (RNAi) Gene Editing Technologies DNA Vaccines Others |
| By End-User | Hospitals Research Institutions Pharmaceutical Companies Biotechnology Firms Others |
| By Application | Cancer Treatment Genetic Disorders Infectious Diseases Cardiovascular Diseases Others |
| By Delivery Method | Viral Vectors Lipid Nanoparticles Electroporation Microinjection Others |
| By Region | Java Sumatra Bali Kalimantan Others |
| By Research Phase | Preclinical Clinical Trials Marketed Products Others |
| By Funding Source | Government Grants Private Investments Venture Capital Corporate Funding Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 150 | Oncologists, Geneticists, Pharmacists |
| Biotechnology Firms | 100 | R&D Managers, Product Development Leads |
| Regulatory Bodies | 50 | Regulatory Affairs Specialists, Policy Makers |
| Patient Advocacy Groups | 80 | Patient Representatives, Healthcare Advocates |
| Clinical Research Organizations | 70 | Clinical Trial Managers, Data Analysts |
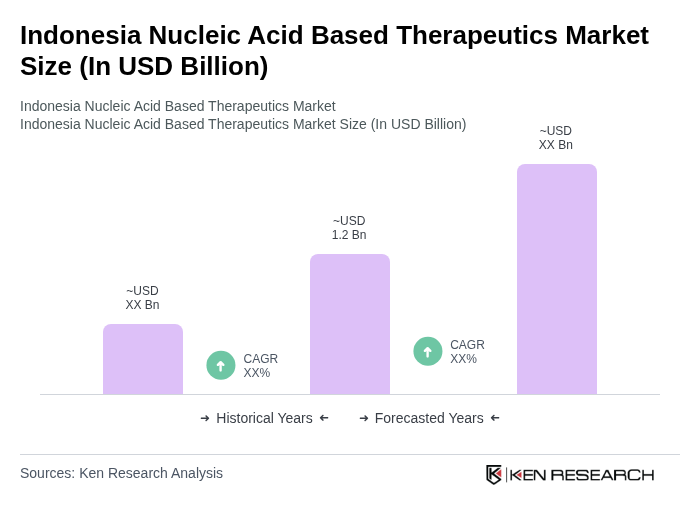
The Indonesia Nucleic Acid Based Therapeutics Market is valued at approximately USD 1.2 billion, reflecting significant growth driven by advancements in biotechnology, increasing genetic disorders, and rising investments in research and development.