Region:Asia
Author(s):Rebecca
Product Code:KRAE2516
Pages:85
Published On:February 2026
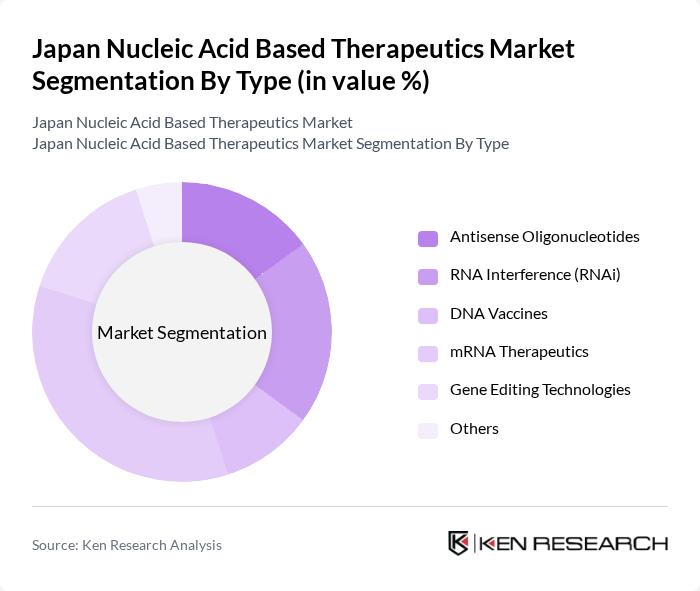
By Type:The market is segmented into various types of nucleic acid-based therapeutics, including Antisense Oligonucleotides, RNA Interference (RNAi), DNA Vaccines, mRNA Therapeutics, Gene Editing Technologies, and Others. Among these, mRNA Therapeutics has gained significant traction due to its pivotal role in vaccine development and therapeutic applications, particularly highlighted during the COVID-19 pandemic. The increasing focus on precision medicine and the ability of mRNA to elicit strong immune responses are driving its adoption in clinical settings.
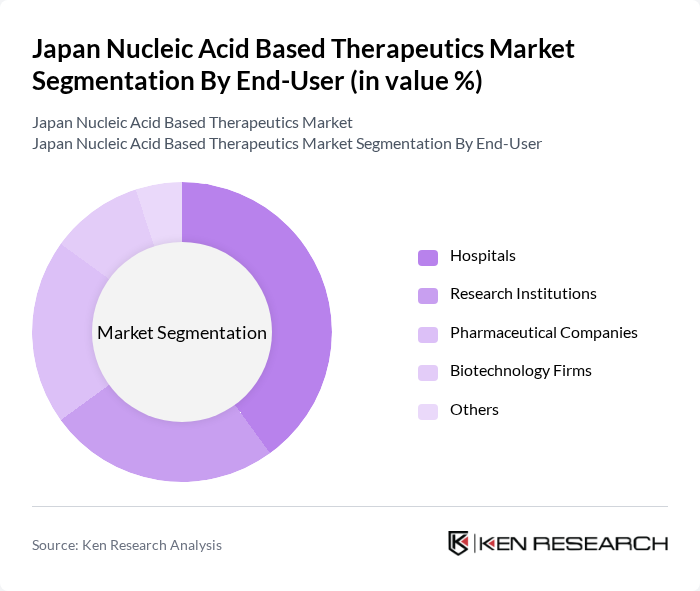
By End-User:The end-user segmentation includes Hospitals, Research Institutions, Pharmaceutical Companies, Biotechnology Firms, and Others. Hospitals are the leading end-users, driven by the increasing adoption of advanced therapeutic options and the need for specialized treatments in clinical settings. The growing collaboration between hospitals and biotech firms for research and development is further enhancing the market dynamics.
The Japan Nucleic Acid Based Therapeutics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Takeda Pharmaceutical Company Limited, Astellas Pharma Inc., Daiichi Sankyo Company, Limited, Chugai Pharmaceutical Co., Ltd., Kyowa Kirin Co., Ltd., Mitsubishi Tanabe Pharma Corporation, Eisai Co., Ltd., Sumitomo Dainippon Pharma Co., Ltd., Sanofi K.K., Pfizer Japan Inc., Amgen K.K., Novartis Pharma K.K., Merck Biopharma K.K., Regeneron Pharmaceuticals, Inc., Gilead Sciences, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the nucleic acid-based therapeutics market in Japan appears promising, driven by ongoing advancements in technology and increasing collaboration among stakeholders. As the healthcare landscape evolves, the integration of artificial intelligence in drug discovery and the expansion of decentralized clinical trials are expected to enhance efficiency and patient engagement. Furthermore, a growing focus on sustainable manufacturing practices will likely shape the industry's operational strategies, ensuring long-term viability and innovation in therapeutic solutions.
| Segment | Sub-Segments |
|---|---|
| By Type | Antisense Oligonucleotides RNA Interference (RNAi) DNA Vaccines mRNA Therapeutics Gene Editing Technologies Others |
| By End-User | Hospitals Research Institutions Pharmaceutical Companies Biotechnology Firms Others |
| By Application | Oncology Genetic Disorders Infectious Diseases Cardiovascular Diseases Others |
| By Delivery Method | Lipid Nanoparticles Viral Vectors Electroporation Microinjection Others |
| By Therapeutic Area | Rare Diseases Autoimmune Disorders Neurological Disorders Metabolic Disorders Others |
| By Region | Kanto Kansai Chubu Kyushu Hokkaido |
| By Research Phase | Preclinical Clinical Trials Marketed Products Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Therapeutics | 100 | Oncologists, Clinical Researchers |
| Genetic Disorder Treatments | 80 | Geneticists, Rare Disease Specialists |
| Infectious Disease Applications | 70 | Infectious Disease Physicians, Pharmacists |
| Regulatory Insights | 50 | Regulatory Affairs Managers, Compliance Officers |
| Patient Advocacy Perspectives | 60 | Patient Advocates, Healthcare Policy Makers |
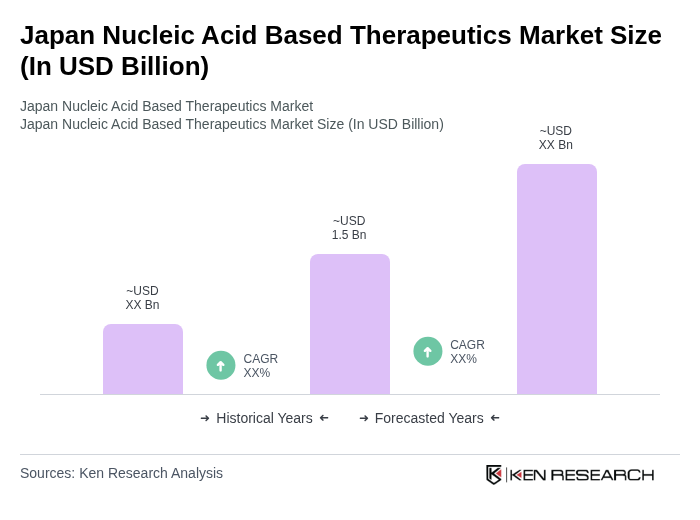
The Japan Nucleic Acid Based Therapeutics Market is valued at approximately USD 1.5 billion, reflecting significant growth driven by advancements in genetic research, increasing prevalence of genetic disorders, and rising investments in biotechnology.