Region:Asia
Author(s):Geetanshi
Product Code:KRAE1136
Pages:100
Published On:February 2026
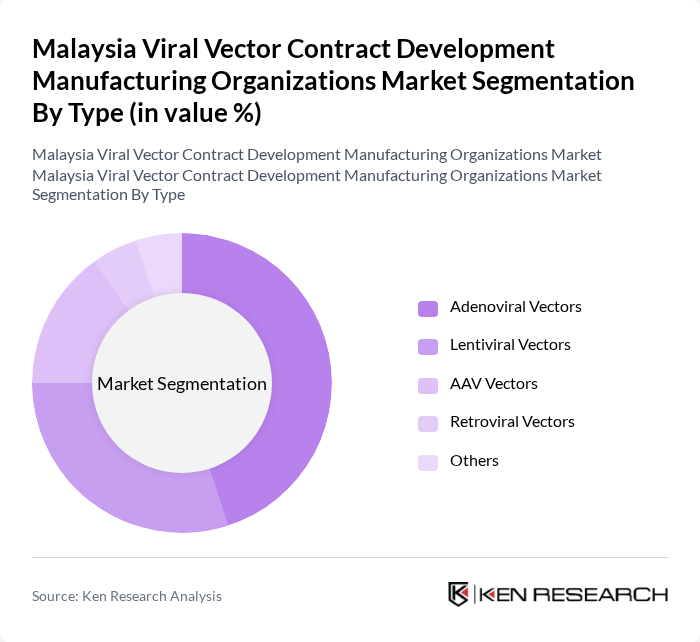
By Type:The market is segmented into various types of viral vectors, including Adenoviral Vectors, Lentiviral Vectors, AAV Vectors, Retroviral Vectors, and Others. Among these, AAV Vectors are currently leading the market due to their favorable safety profiles, tissue tropism, and multiple approved therapies, making them ideal for gene therapy applications. Lentiviral Vectors are also gaining traction, particularly in the field of gene editing and cell therapy, driven by their stable integration into the host genome.
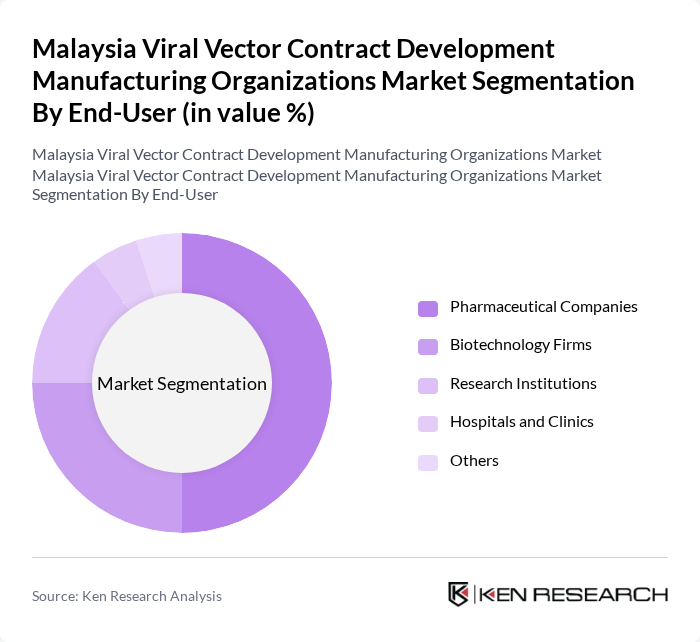
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Research Institutions, Hospitals and Clinics, and Others. Pharmaceutical Companies dominate this segment due to their extensive resources and capabilities in developing and commercializing viral vector-based therapies. Biotechnology Firms are also significant players, focusing on innovative solutions and niche markets, while Research Institutions contribute to the foundational research that drives advancements in this field.
The Malaysia Viral Vector Contract Development Manufacturing Organizations Market is characterized by a dynamic mix of regional and international players. Leading participants such as Biocon, GenScript, Lonza, WuXi AppTec, Merck KGaA, Thermo Fisher Scientific, Catalent, AAVLife, Oxford Biomedica, Virovek, Astellas Pharma, Takeda Pharmaceutical Company, Amgen, GSK, and Novartis contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Malaysia viral vector contract development manufacturing organizations market appears promising, driven by increasing collaboration between academia and industry. As the demand for personalized medicine grows, the focus on tailored gene therapies will likely expand. Furthermore, advancements in artificial intelligence and machine learning are expected to streamline development processes, enhancing efficiency and reducing costs. These trends indicate a vibrant market landscape, fostering innovation and attracting investment in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Adenoviral Vectors Lentiviral Vectors AAV Vectors Retroviral Vectors Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Research Institutions Hospitals and Clinics Others |
| By Application | Gene Therapy Vaccine Development Oncolytic Therapy Others |
| By Delivery Method | In Vivo Delivery Ex Vivo Delivery Others |
| By Production Scale | Preclinical Scale Clinical Scale Commercial Scale Others |
| By Region | Central Region Northern Region Southern Region Eastern Region |
| By Regulatory Compliance | Local Regulations International Standards Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Viral Vector Manufacturing Services | 120 | Production Managers, Quality Assurance Leads |
| Gene Therapy Development | 100 | R&D Directors, Clinical Project Managers |
| Regulatory Compliance in Biotech | 80 | Regulatory Affairs Specialists, Compliance Officers |
| Market Trends in Biomanufacturing | 90 | Market Analysts, Business Development Managers |
| Investment in Biotech Startups | 60 | Venture Capitalists, Angel Investors |
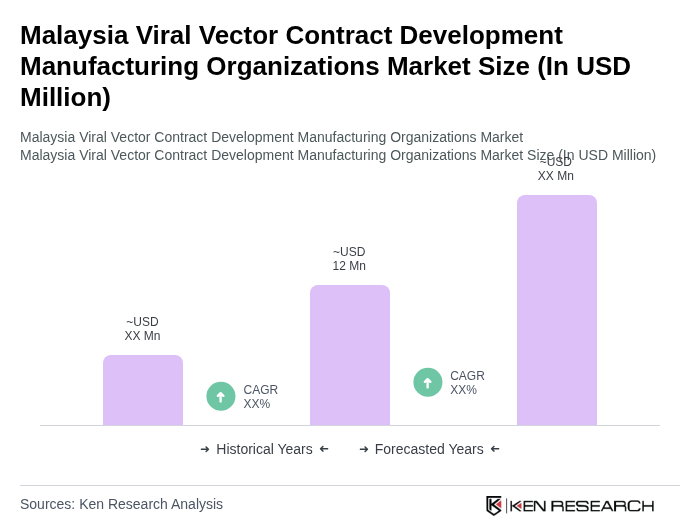
The Malaysia Viral Vector Contract Development Manufacturing Organizations market is valued at approximately USD 12 million, driven by the increasing demand for advanced therapies, particularly in gene therapy and vaccine development, as well as the rising prevalence of chronic diseases.