Region:Asia
Author(s):Geetanshi
Product Code:KRAE1143
Pages:85
Published On:February 2026
By Vector Type:
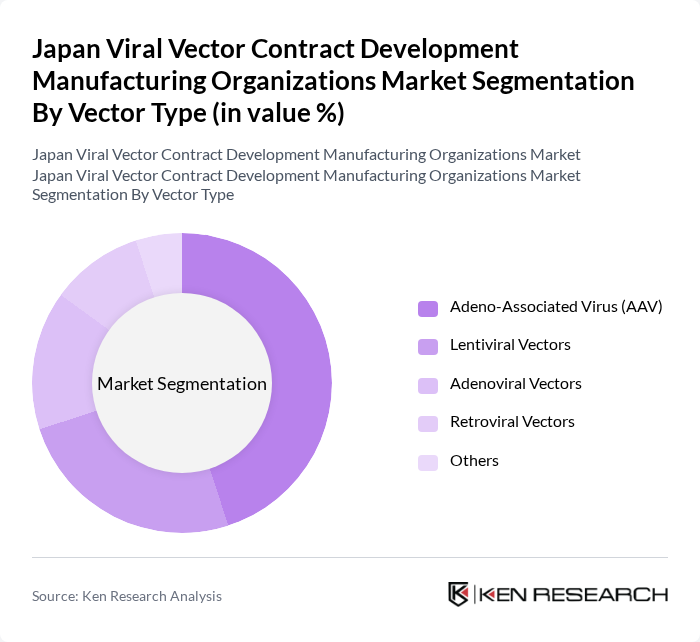
The segmentation by vector type includes Adeno-Associated Virus (AAV), Lentiviral Vectors, Adenoviral Vectors, Retroviral Vectors, and Others. Among these, Adeno-Associated Virus (AAV) is the leading subsegment, primarily due to its favorable safety profile and efficiency in gene delivery. AAV vectors are widely used in gene therapy applications, which have seen a surge in demand as more therapies receive regulatory approval. The versatility of AAV in targeting various tissues and its ability to induce long-term expression of therapeutic genes further solidify its dominance in the market.
By End-User:
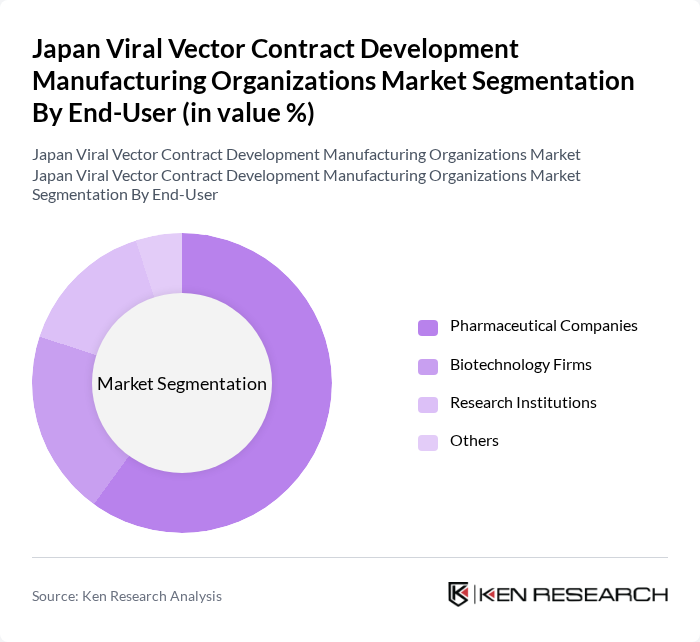
This segmentation includes Pharmaceutical Companies, Biotechnology Firms, Research Institutions, and Others. Pharmaceutical Companies dominate this segment, driven by their extensive resources and capabilities in developing and commercializing viral vector-based therapies. These companies are increasingly investing in R&D to innovate and expand their product pipelines, particularly in gene therapy and vaccine development. The collaboration with biotechnology firms and research institutions further enhances their ability to leverage cutting-edge technologies and accelerate the development of new therapies.
The Japan Viral Vector Contract Development Manufacturing Organizations Market is characterized by a dynamic mix of regional and international players. Leading participants such as Takara Bio Inc., Astellas Pharma Inc., Fujifilm Diosynth Biotechnologies, JCR Pharmaceuticals Co., Ltd., Daiichi Sankyo Company, Limited, Chugai Pharmaceutical Co., Ltd., Kyowa Kirin Co., Ltd., Sanofi K.K., Mitsubishi Tanabe Pharma Corporation, Sumitomo Dainippon Pharma Co., Ltd., Gene Therapy Research Institute, Medicago Inc., AAVLife, OncoOne, Virovek, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the viral vector CDMO market in Japan appears promising, driven by ongoing advancements in technology and increasing collaboration between academia and industry. As the demand for personalized medicine grows, CDMOs are likely to adapt their services to meet these needs. Furthermore, the focus on rare diseases is expected to create new avenues for growth, as innovative therapies emerge to address previously unmet medical needs, enhancing the overall market landscape.
| Segment | Sub-Segments |
|---|---|
| By Vector Type | Adeno-Associated Virus (AAV) Lentiviral Vectors Adenoviral Vectors Retroviral Vectors Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Research Institutions Others |
| By Application | Gene Therapy Vaccine Development Oncolytic Therapy Others |
| By Delivery Method | In Vivo Delivery Ex Vivo Delivery Others |
| By Manufacturing Process | Transient Transfection Stable Cell Line Others |
| By Region | Kanto Kansai Chubu Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gene Therapy Development | 45 | R&D Directors, Project Managers |
| Vaccine Production Services | 40 | Production Managers, Quality Assurance Leads |
| Regulatory Compliance in Biotech | 40 | Regulatory Affairs Managers, Compliance Officers |
| Market Trends in Viral Vectors | 50 | Market Analysts, Business Development Executives |
| Contract Manufacturing Insights | 45 | Operations Managers, Strategic Planners |
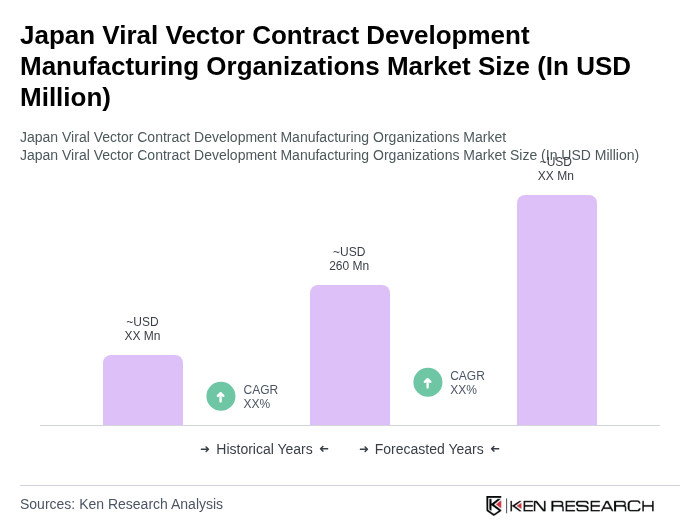
The Japan Viral Vector Contract Development Manufacturing Organizations market is valued at approximately USD 260 million, driven by the increasing demand for gene therapies and vaccines, advancements in biotechnology, and rising clinical trial activities.