Region:Asia
Author(s):Geetanshi
Product Code:KRAE1137
Pages:93
Published On:February 2026
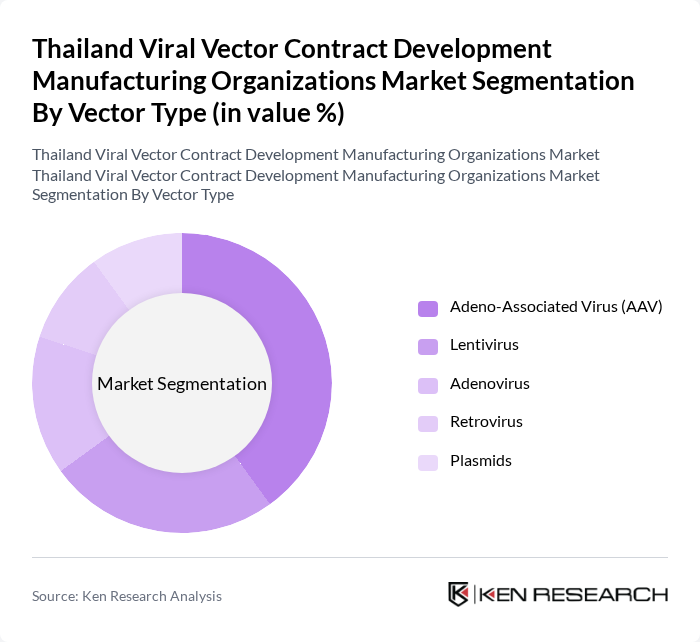
By Vector Type:The market is segmented into various vector types, including Adeno-Associated Virus (AAV), Lentivirus, Adenovirus, Retrovirus, and Plasmids. Among these, Adeno-Associated Virus (AAV) is currently the leading subsegment due to its favorable safety profile and efficiency in gene delivery, making it a preferred choice for therapeutic applications. The increasing number of clinical trials utilizing AAV vectors is driving its dominance in the market.
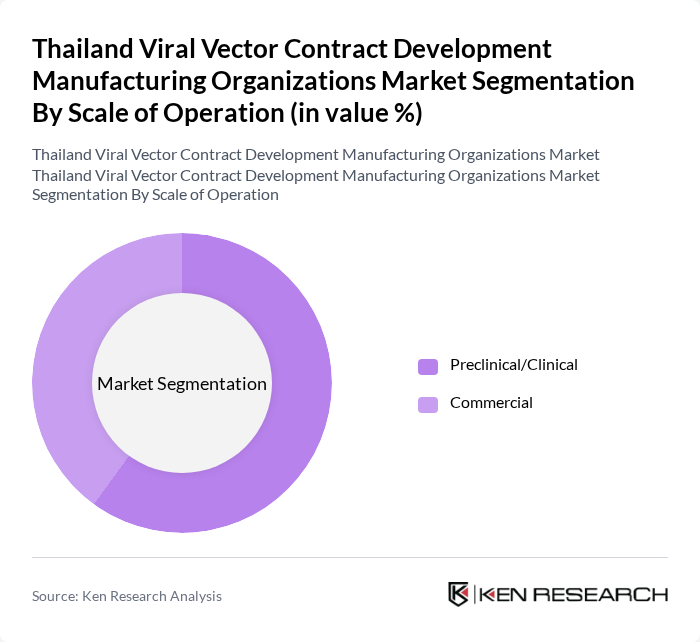
By Scale of Operation:The market is divided into Preclinical/Clinical and Commercial scales of operation. The Preclinical/Clinical segment is currently leading the market, driven by the surge in research activities and clinical trials for gene therapies and vaccines. The increasing investment in R&D by pharmaceutical and biotechnology companies is propelling this segment's growth, as it allows for the exploration of innovative treatment options.
The Thailand Viral Vector Contract Development Manufacturing Organizations Market is characterized by a dynamic mix of regional and international players. Leading participants such as Siam Bioscience, BioNet-Asia, GPO (Government Pharmaceutical Organization), Chulalongkorn University, Biocure Pharmaceutical, Genexine, Takeda Pharmaceutical, Asembia, ViroMed, Medigen Vaccine Biologics Corp., Thonburi Healthcare Group, Bangkok Biomed, Biopharma, TPI Polene, Thai Biopharma contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Thailand viral vector contract development manufacturing organizations market appears promising, driven by increasing collaborations between public and private sectors. As the demand for personalized medicine rises, organizations are likely to focus on developing tailored therapies. Furthermore, advancements in technology, particularly in artificial intelligence and automation, are expected to streamline production processes, enhancing efficiency and reducing costs. This evolving landscape will create a fertile ground for innovation and growth in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Vector Type | Adeno-Associated Virus (AAV) Lentivirus Adenovirus Retrovirus Plasmids |
| By Scale of Operation | Preclinical/Clinical Commercial |
| By Workflow | Upstream Processing Downstream Processing |
| By Delivery Method | In Vivo Ex Vivo |
| By End-User | Pharmaceutical Companies Biotechnology Companies Contract Research Organizations Contract Development and Manufacturing Organizations Academic/Research Institutes |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Viral Vector Production Services | 100 | Production Managers, Quality Assurance Leads |
| Gene Therapy Development | 80 | R&D Directors, Clinical Trial Managers |
| Vaccine Manufacturing Partnerships | 70 | Business Development Executives, Regulatory Affairs Specialists |
| Contract Research Services | 60 | Project Managers, Scientific Advisors |
| Market Entry Strategies for Biotech | 90 | Market Analysts, Strategy Consultants |
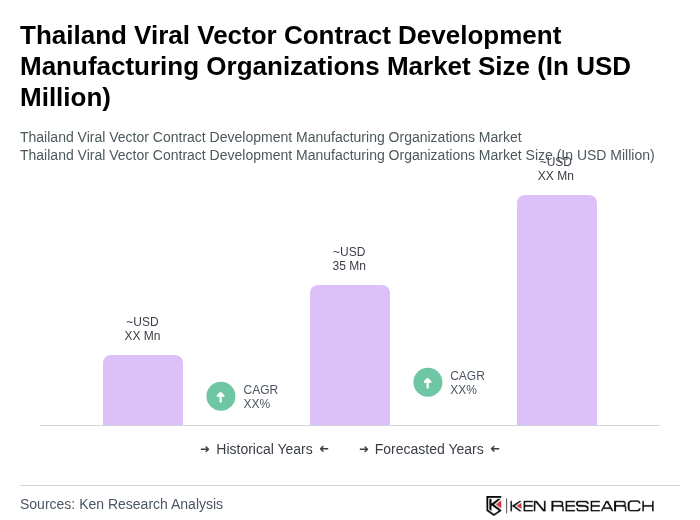
The Thailand Viral Vector Contract Development Manufacturing Organizations market is valued at approximately USD 35 million, reflecting a significant growth driven by the rising demand for gene therapies and vaccines, particularly in response to the COVID-19 pandemic.