Region:Global
Author(s):Geetanshi
Product Code:KRAE1144
Pages:88
Published On:February 2026
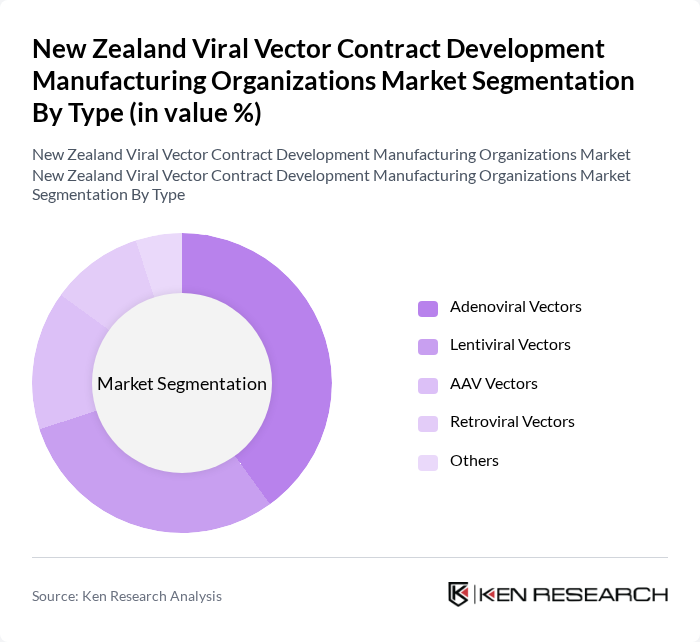
By Type:The market can be segmented into various types of viral vectors, including Adenoviral Vectors, Lentiviral Vectors, AAV Vectors, Retroviral Vectors, and Others. Among these, AAV Vectors are currently leading the market due to their favorable safety profiles and multiple approved therapies, making them ideal for gene therapy applications. Lentiviral Vectors are also gaining traction, particularly in the field of gene editing and cell therapy, as they can transduce both dividing and non-dividing cells, providing long-term expression of therapeutic genes.
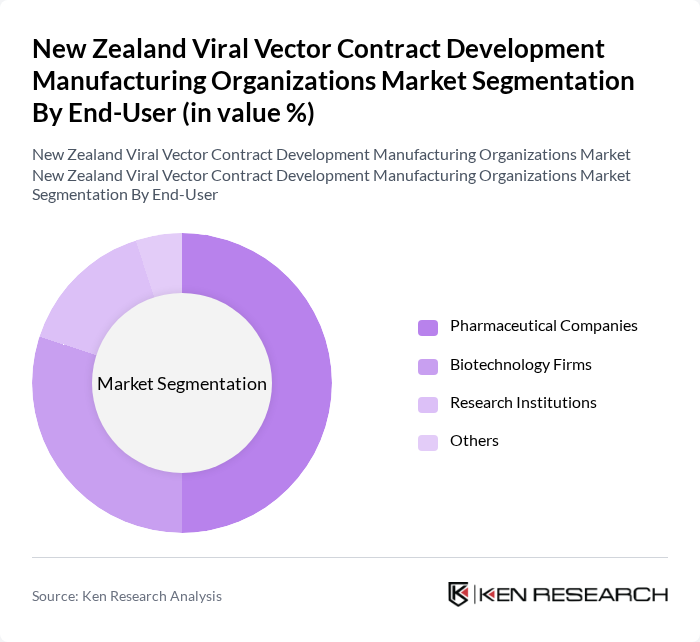
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Research Institutions, and Others. Pharmaceutical Companies dominate the market as they invest heavily in R&D for developing novel therapies, particularly in gene therapy and vaccine production. Biotechnology Firms are also significant players, focusing on innovative solutions and collaborations with research institutions to advance viral vector technologies.
The New Zealand Viral Vector Contract Development Manufacturing Organizations Market is characterized by a dynamic mix of regional and international players. Leading participants such as AFT Pharmaceuticals, Fisher & Paykel Healthcare, Genentech, Medsafe, Auckland Bioengineering Institute, Wellington Institute of Technology, Christchurch School of Medicine, Victoria University of Wellington, University of Otago, AgResearch, Callaghan Innovation, Bioculture, Southern Cross University, BioPacific Partners, NZ Bio contribute to innovation, geographic expansion, and service delivery in this space.
The future of the New Zealand viral vector contract development manufacturing organizations market appears promising, driven by ongoing advancements in gene therapy technologies and increasing collaboration between research institutions and industry players. As the demand for personalized medicine grows, organizations are likely to focus on developing tailored therapies that address specific genetic disorders. Additionally, the integration of artificial intelligence in manufacturing processes is expected to enhance efficiency and reduce costs, further propelling market growth in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Adenoviral Vectors Lentiviral Vectors AAV Vectors Retroviral Vectors Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Research Institutions Others |
| By Application | Gene Therapy Vaccine Development Oncolytic Virus Therapy Others |
| By Delivery Method | In Vivo Delivery Ex Vivo Delivery Others |
| By Manufacturing Scale | Small Scale Medium Scale Large Scale Others |
| By Region | North Island South Island |
| By Regulatory Compliance | FDA Compliance EMA Compliance Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Viral Vector Manufacturing Services | 100 | Operations Managers, Production Directors |
| Gene Therapy Development | 80 | Research Scientists, Clinical Development Managers |
| Vaccine Production Facilities | 70 | Quality Control Managers, Regulatory Affairs Specialists |
| Biotech Startups Utilizing Viral Vectors | 60 | Founders, Chief Technology Officers |
| Academic Institutions Conducting Viral Vector Research | 50 | Principal Investigators, Lab Managers |
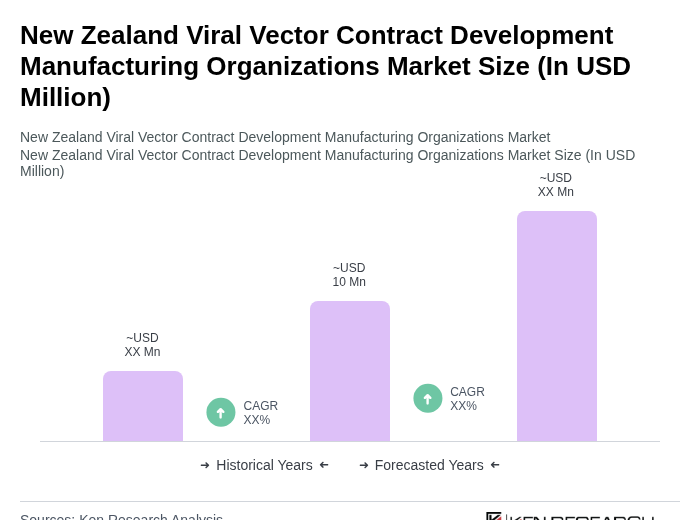
The New Zealand Viral Vector Contract Development Manufacturing Organizations Market is valued at approximately USD 10 million, reflecting a growing demand for advanced therapies, particularly in gene therapy and vaccine development, driven by the rising prevalence of genetic disorders and infectious diseases.