Region:Asia
Author(s):Geetanshi
Product Code:KRAE1142
Pages:82
Published On:February 2026
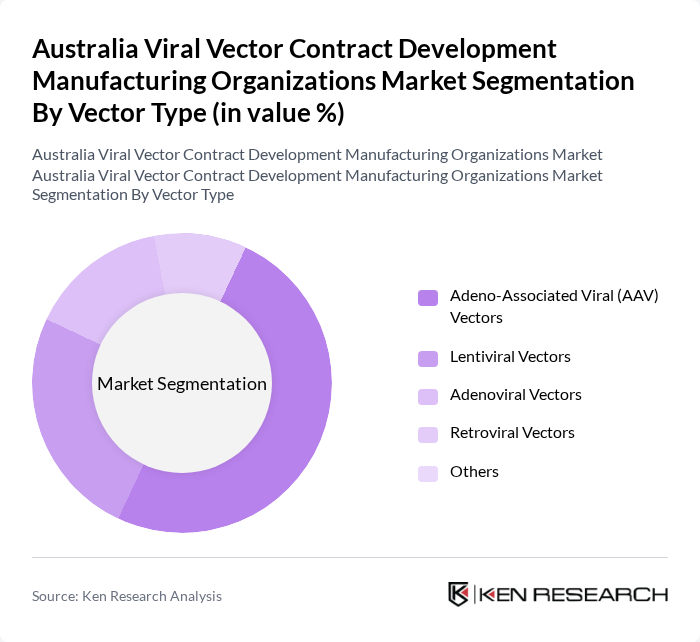
By Vector Type:The viral vector market can be segmented into various vector types, including Adeno-Associated Viral (AAV) Vectors, Lentiviral Vectors, Adenoviral Vectors, Retroviral Vectors, and Others. Among these, Adeno-Associated Viral (AAV) Vectors are currently leading the market due to their favorable safety profile and efficiency in gene delivery. The increasing adoption of AAV vectors in clinical trials for gene therapies is a significant factor driving their dominance.
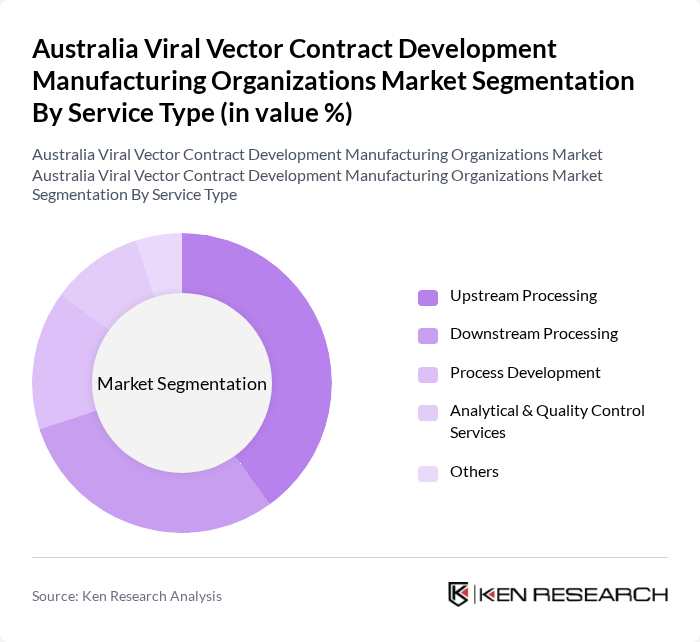
By Service Type:The service type segmentation includes Upstream Processing, Downstream Processing, Process Development, Analytical & Quality Control Services, and Others. Upstream Processing is the leading segment, driven by the increasing need for efficient production processes in the development of viral vectors. The focus on optimizing cell culture and vector production techniques has made this segment crucial for manufacturers.
The Australia Viral Vector Contract Development Manufacturing Organizations Market is characterized by a dynamic mix of regional and international players. Leading participants such as Viral Vector Manufacturing Facility (VVMF), CSL Limited, Mesoblast Limited, Imugene Limited, Clinuvel Pharmaceuticals Ltd, Cynata Therapeutics Ltd contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Australia viral vector contract development manufacturing organizations market appears promising, driven by ongoing advancements in gene therapy and biotechnology. As the demand for personalized medicine grows, companies are likely to invest in innovative manufacturing technologies to enhance efficiency. Additionally, collaborations between research institutions and biotech firms are expected to increase, fostering a more dynamic environment for the development of cutting-edge therapies. This collaborative approach will likely lead to accelerated product development timelines and improved patient outcomes.
| Segment | Sub-Segments |
|---|---|
| By Vector Type | Adeno-Associated Viral (AAV) Vectors Lentiviral Vectors Adenoviral Vectors Retroviral Vectors Others |
| By Service Type | Upstream Processing Downstream Processing Process Development Analytical & Quality Control Services Others |
| By Application | Gene Therapy Cell Therapy Vaccine Development Oncolytic Virotherapy Others |
| By Scale of Operation | Preclinical/Research Grade Clinical (Phase I/II) Commercial Scale |
| By End-User | Biotech Companies Pharmaceutical Companies Academic & Research Institutions Others |
| By Region | New South Wales Victoria Queensland Western Australia Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gene Therapy Development | 100 | R&D Directors, Project Managers |
| Vaccine Manufacturing Services | 80 | Production Managers, Quality Control Specialists |
| Regulatory Compliance in Biotech | 60 | Regulatory Affairs Managers, Compliance Officers |
| Market Trends in Viral Vectors | 90 | Market Analysts, Business Development Executives |
| Investment in Biotech Startups | 70 | Venture Capitalists, Financial Analysts |
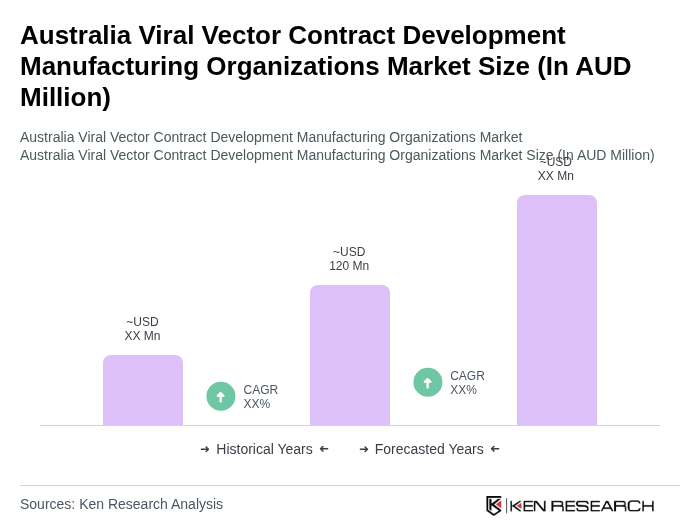
The Australia Viral Vector Contract Development Manufacturing Organizations market is valued at approximately AUD 120 million, driven by the increasing demand for advanced therapies, particularly in gene and cell therapy, and the rising prevalence of genetic disorders and cancers.