Region:Asia
Author(s):Geetanshi
Product Code:KRAE1138
Pages:96
Published On:February 2026
By Type:The market is segmented into various types of viral vectors, including Adenoviral Vectors, Lentiviral Vectors, AAV Vectors, Retroviral Vectors, and Others. Among these, AAV Vectors are currently leading the market due to their favorable safety profiles, high efficiency in gene delivery, and multiple approved therapies in gene therapy and vaccine development. The increasing focus on gene therapy, personalized medicine, and in-vivo applications is further propelling the demand for these vectors.

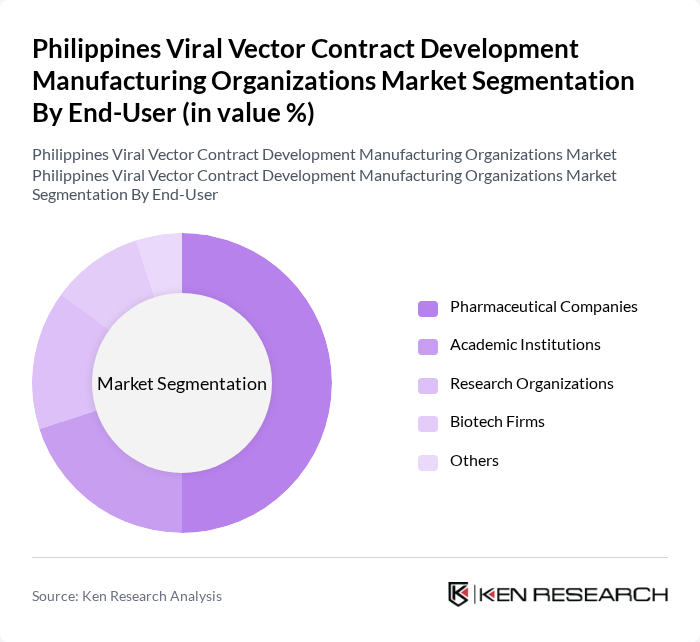
By End-User:The end-user segmentation includes Pharmaceutical Companies, Academic Institutions, Research Organizations, Biotech Firms, and Others. Pharmaceutical Companies dominate this segment due to their extensive investment in R&D for new therapies and vaccines, which increasingly rely on viral vectors for effective delivery mechanisms.
The Philippines Viral Vector Contract Development Manufacturing Organizations Market is characterized by a dynamic mix of regional and international players. Leading participants such as Philippine Genome Center, BioPharma Solutions, Genetica, ViroMed, Philippine Viral Vector Technologies, GeneTech Philippines, Biovector Solutions, Vector Biopharma, GeneVax, Viral Vector Innovations, MedVax, VectorGen, BioVector Labs, GeneCraft, VectorWorks contribute to innovation, geographic expansion, and service delivery in this space.
The future of the viral vector contract development manufacturing sector in the Philippines appears promising, driven by increasing investments and technological advancements. As the demand for gene therapies continues to rise, local manufacturers are expected to enhance their production capabilities. Additionally, collaborations with global biotech firms will likely facilitate knowledge transfer and innovation. The integration of automation and digital technologies will further streamline operations, positioning the Philippines as a competitive player in the global market for viral vector applications.
| Segment | Sub-Segments |
|---|---|
| By Type | Adenoviral Vectors Lentiviral Vectors AAV Vectors Retroviral Vectors Others |
| By End-User | Pharmaceutical Companies Academic Institutions Research Organizations Biotech Firms Others |
| By Application | Gene Therapy Vaccine Development Oncolytic Virus Therapy Others |
| By Delivery Method | In Vivo Delivery Ex Vivo Delivery Others |
| By Manufacturing Process | Upstream Processing Downstream Processing Others |
| By Scale of Production | Small Scale Medium Scale Large Scale Others |
| By Geographic Focus | Metro Manila Luzon Visayas Mindanao Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Viral Vector Manufacturing Services | 100 | Operations Managers, Production Directors |
| Gene Therapy Development | 80 | Research Scientists, Clinical Development Managers |
| Vaccine Production Capabilities | 70 | Regulatory Affairs Specialists, Quality Assurance Managers |
| Market Trends in Biotechnology | 90 | Market Analysts, Business Development Executives |
| Investment in Biotech Startups | 60 | Venture Capitalists, Industry Consultants |
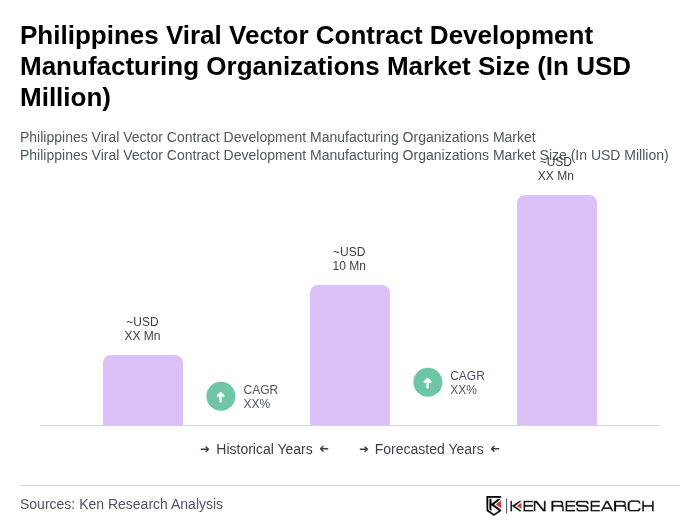
The Philippines Viral Vector Contract Development Manufacturing Organizations market is valued at approximately USD 10 million, driven by the increasing demand for advanced therapies, particularly in gene therapy and vaccine development, alongside a rise in R&D activities in the biotechnology sector.