Region:Middle East
Author(s):Shubham
Product Code:KRAD2446
Pages:87
Published On:January 2026
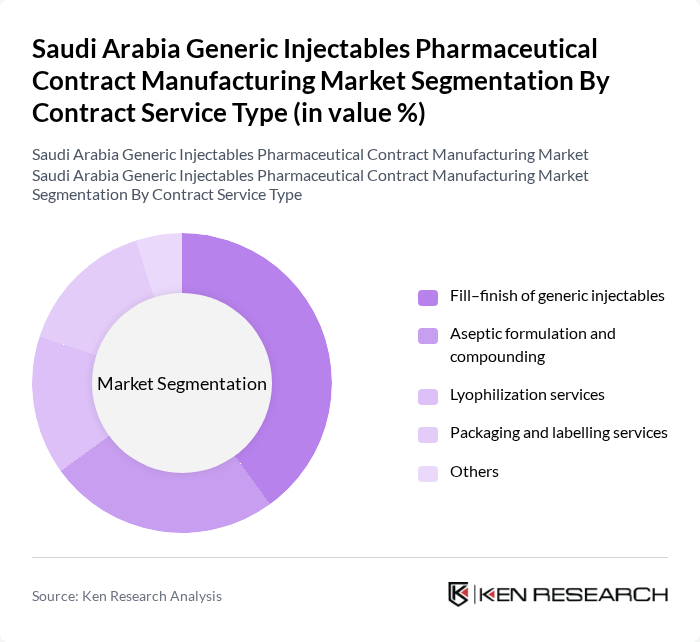
By Contract Service Type:The contract service type segmentation includes various services that cater to the needs of pharmaceutical companies. The dominant sub-segment is the fill-finish of generic injectables, which is crucial for ensuring the safe and effective delivery of medications and is a core focus area for Saudi-based parenteral manufacturers expanding capacity under localization initiatives. Aseptic formulation and compounding also play a significant role, especially in the production of sterile injectables for high-volume therapeutic areas such as anti-infectives, analgesics, and cardiovascular drugs. Lyophilization services are gaining traction due to the increasing demand for stable formulations for biologics and sensitive molecules, while packaging and labeling services are essential for compliance with SFDA requirements, serialization, and marketability. The 'Others' category includes additional services that support the manufacturing process, such as analytical testing, stability studies, and technology transfer support.
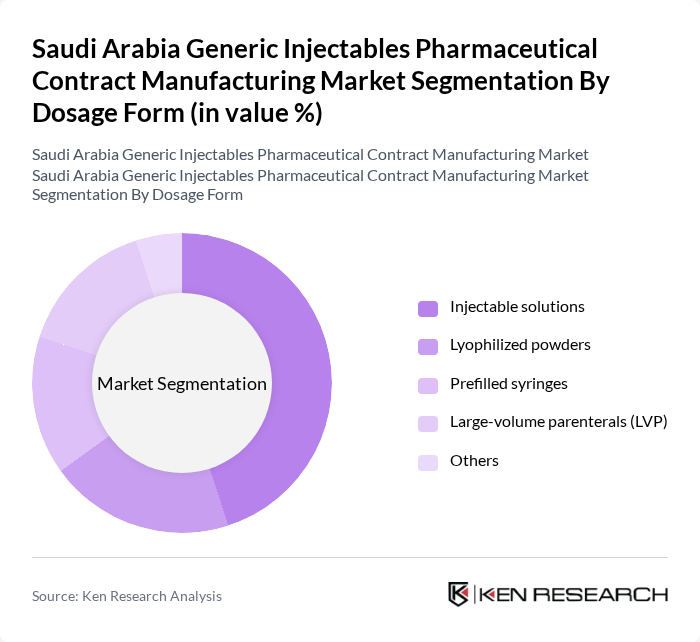
By Dosage Form:The dosage form segmentation encompasses various types of injectable products. Injectable solutions are the most widely used due to their ease of administration, suitability for high-volume hospital use, and rapid onset of action in emergency and critical care settings. Lyophilized powders are also significant, particularly for biologics and sensitive compounds that require enhanced stability and reconstitution at the point of care. Prefilled syringes are gaining popularity for their convenience, reduced risk of contamination, and support for self-administration in chronic disease management. Large-volume parenterals (LVP) are essential for delivering fluids, electrolytes, nutrition, and certain drugs, especially in hospital and intensive care settings. The 'Others' category includes various specialized dosage forms, such as cartridges and dual-chamber systems, that cater to niche markets and more advanced therapeutic applications.
The Saudi Arabia Generic Injectables Pharmaceutical Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Saudi Pharmaceutical Industries and Medical Appliances Corporation (SPIMACO), Tabuk Pharmaceuticals Manufacturing Company, Jamjoom Pharmaceuticals Company, Riyadh Pharma (Medical and Pharmaceutical Services Co.), Banaja Holding Company, Tamer Group, Sudair Pharma Company, Jamjoom Medical Industries, Gulf Pharmaceutical Industries (Julphar), Aurobindo Pharma / local JV entities, Hikma Pharmaceuticals / local operations, SPIMACO Addwaeih / contract manufacturing subsidiaries, Arabio (Saudi Arabian Japanese Pharmaceutical Company), Saudi Chemical Company Holding – Pharma segment, Dar Al Dawa Development and Investment Company contribute to innovation, geographic expansion, and service delivery in this space through investments in sterile injectable facilities, local partnerships, and government-tender focused portfolios.
The future of the Saudi Arabia generic injectables market appears promising, driven by ongoing investments in healthcare infrastructure and a growing emphasis on local manufacturing. As the government continues to support initiatives aimed at enhancing domestic production capabilities, local manufacturers are likely to see increased opportunities for growth. Additionally, the rising prevalence of chronic diseases will further fuel demand for affordable healthcare solutions, positioning the market for significant expansion in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Contract Service Type | Fill–finish of generic injectables Aseptic formulation and compounding Lyophilization services Packaging and labelling services Others |
| By Dosage Form | Injectable solutions Lyophilized powders Prefilled syringes Large-volume parenterals (LVP) Others |
| By Therapeutic Area | Oncology Anti-infectives Cardiovascular Anesthetics and pain management Parenteral nutrition Others |
| By Client Type | Multinational pharmaceutical companies Regional and local generic companies Biotech and specialty pharma Government and institutional clients Others |
| By Packaging Container | Vials Ampoules Cartridges Prefilled syringes Others |
| By Manufacturing Scale | Clinical-scale manufacturing Commercial-scale manufacturing Small-batch / on-demand manufacturing |
| By Region | Northern and Central Region Western Region Eastern Region Southern Region |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 100 | CEOs, Production Managers |
| Healthcare Providers | 90 | Pharmacy Directors, Procurement Officers |
| Regulatory Bodies | 60 | Regulatory Affairs Specialists, Compliance Officers |
| Market Analysts | 70 | Market Research Analysts, Industry Consultants |
| Healthcare Policy Makers | 50 | Health Economists, Policy Advisors |
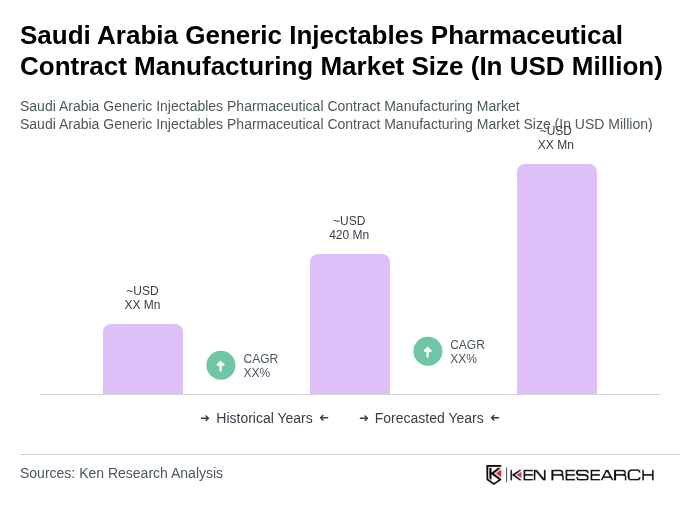
The Saudi Arabia Generic Injectables Pharmaceutical Contract Manufacturing Market is valued at approximately USD 420 million, reflecting a significant growth driven by increasing demand for affordable healthcare solutions and the rise in chronic diseases.