Region:North America
Author(s):Rebecca
Product Code:KRAB0879
Pages:82
Published On:December 2025
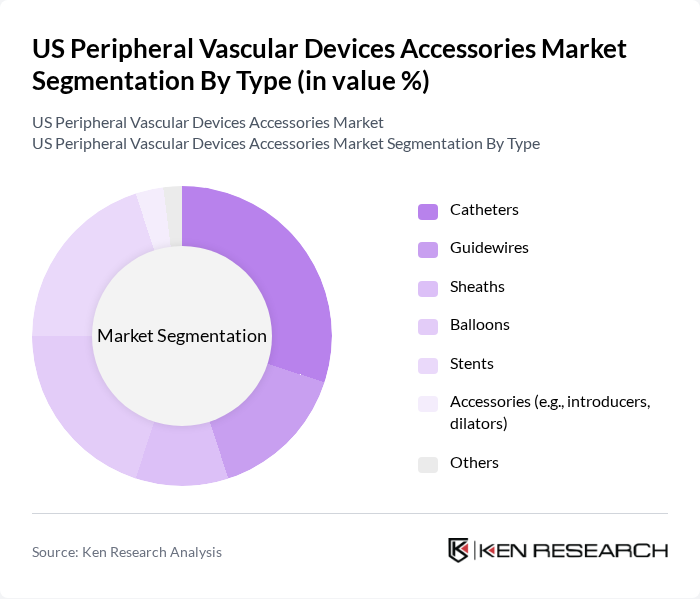
By Type:The market is segmented into various types of accessories, including catheters, guidewires, sheaths, balloons, stents, and other accessories such as introducers and dilators. Among these, catheters and stents are the most prominent due to their critical role in interventional procedures. The increasing adoption of minimally invasive techniques has led to a surge in demand for these devices, as they offer improved patient outcomes and reduced recovery times.
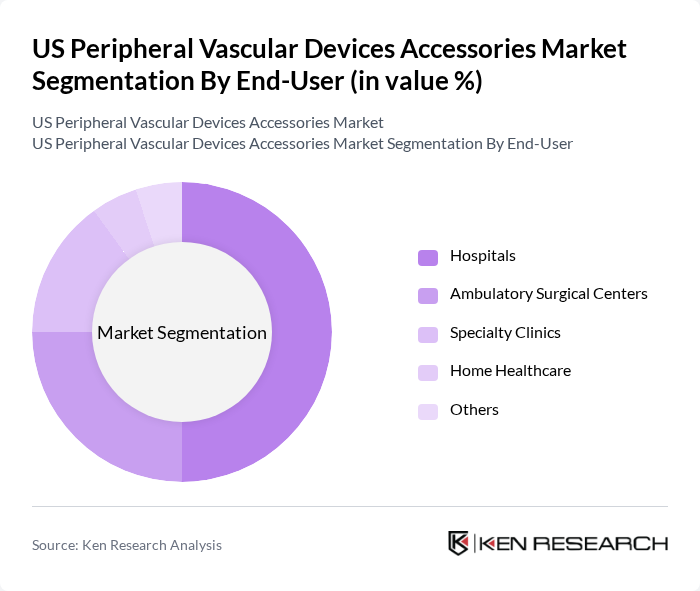
By End-User:The end-user segmentation includes hospitals, ambulatory surgical centers, specialty clinics, home healthcare, and others. Hospitals are the leading end-users, primarily due to their capacity to perform complex vascular procedures and the availability of advanced medical technologies. The trend towards outpatient procedures is also driving growth in ambulatory surgical centers, which are increasingly preferred for their efficiency and cost-effectiveness.
The US Peripheral Vascular Devices Accessories Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Boston Scientific, Abbott Laboratories, Terumo Corporation, B. Braun Melsungen AG, Cook Medical, Cardinal Health, Johnson & Johnson, Philips Healthcare, Gore Medical, Merit Medical Systems, Siemens Healthineers, Stryker Corporation, Edwards Lifesciences, Hologic, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the US Peripheral Vascular Devices Accessories Market appears promising, driven by ongoing technological innovations and a demographic shift towards an aging population. The integration of digital health technologies and artificial intelligence is expected to enhance patient care and operational efficiency. Additionally, the market is likely to see increased consolidation through mergers and acquisitions, allowing companies to offer comprehensive solutions that combine devices and accessories, ultimately improving patient outcomes and market competitiveness.
| Segment | Sub-Segments |
|---|---|
| By Type | Catheters Guidewires Sheaths Balloons Stents Accessories (e.g., introducers, dilators) Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Home Healthcare Others |
| By Application | Interventional Cardiology Interventional Radiology Peripheral Vascular Interventions Others |
| By Material | Polyurethane Silicone Polyethylene Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Northeast Midwest South West |
| By Product Lifecycle Stage | Introduction Growth Maturity Decline Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Vascular Surgeons | 100 | Interventional Cardiologists, Vascular Surgeons |
| Hospital Procurement Managers | 80 | Supply Chain Managers, Purchasing Directors |
| Medical Device Sales Representatives | 70 | Sales Managers, Territory Representatives |
| Healthcare Policy Experts | 50 | Health Economists, Policy Analysts |
| Clinical Researchers | 60 | Clinical Trial Coordinators, Research Scientists |
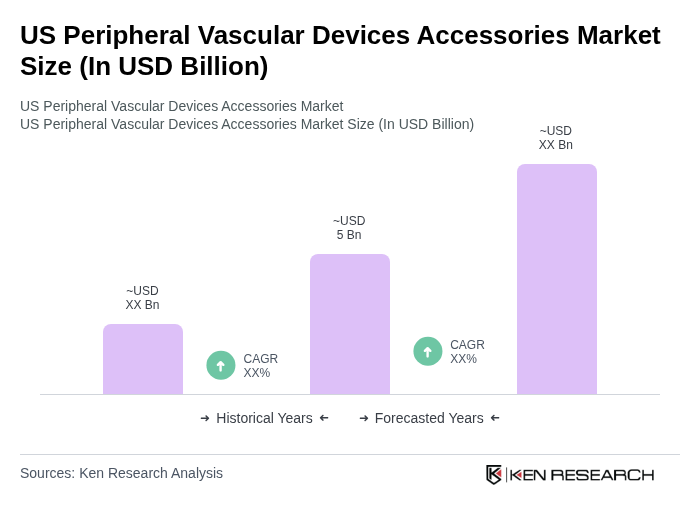
The US Peripheral Vascular Devices Accessories Market is valued at approximately USD 5 billion, reflecting a significant growth driven by factors such as the rising prevalence of peripheral arterial disease (PAD) and advancements in medical technologies.