Region:Asia
Author(s):Geetanshi
Product Code:KRAE1966
Pages:99
Published On:February 2026
By Product:The product segmentation includes various forms of transdermal drug delivery systems, which are essential for effective medication administration. The subsegments are Transdermal Patches, Transdermal Gels, Transdermal Sprays, and Others. Each of these subsegments caters to different therapeutic needs and patient preferences, influencing their market performance.
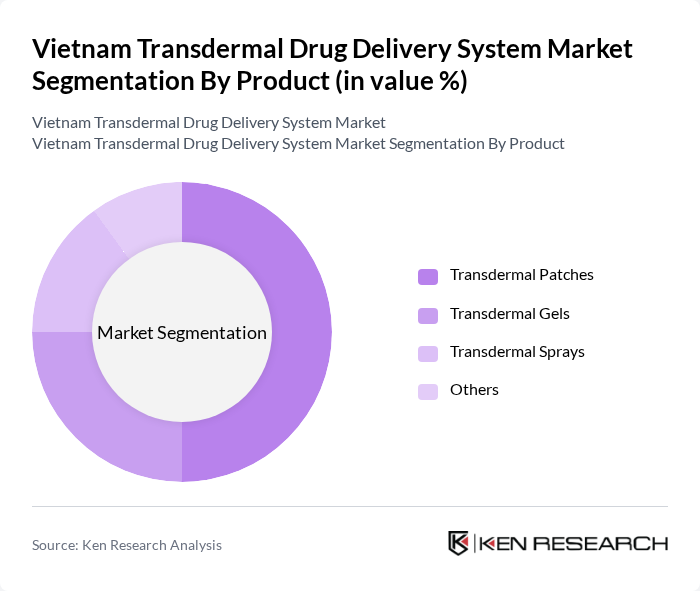
The Transdermal Patches subsegment is currently dominating the market due to their ease of use, effectiveness in delivering medication over extended periods, and patient compliance. These patches are particularly popular for pain management and hormonal therapies, as they provide a steady release of medication without the need for injections or frequent dosing. The convenience and non-invasive nature of patches have led to increased adoption among patients and healthcare providers alike.
By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgery Centers, Home Care Settings, and Others. Each of these segments plays a crucial role in the distribution and utilization of transdermal drug delivery systems, reflecting the diverse applications and settings in which these products are used.
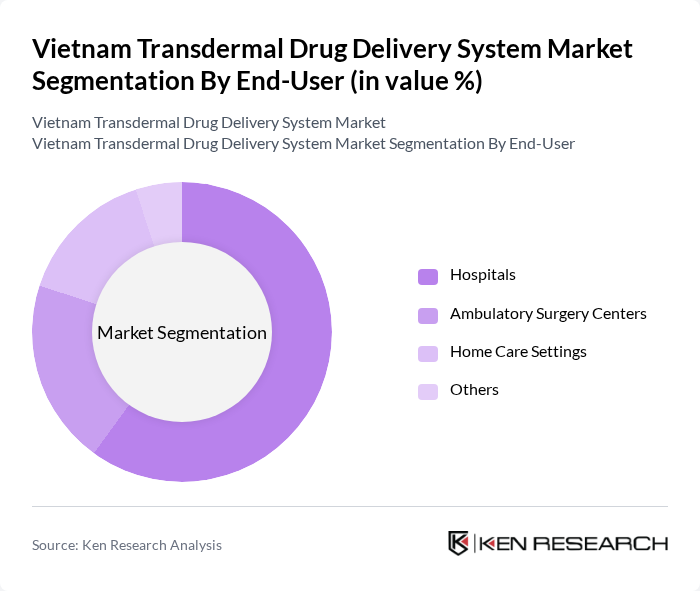
Hospitals are the leading end-user segment, accounting for a significant share of the market. This dominance is attributed to the high volume of patients requiring medication for chronic conditions, the availability of advanced healthcare facilities, and the preference for transdermal systems in acute care settings. Hospitals also benefit from established relationships with pharmaceutical companies, ensuring a steady supply of transdermal products.
The Vietnam Transdermal Drug Delivery System Market is characterized by a dynamic mix of regional and international players. Leading participants such as Johnson & Johnson, Novartis AG, Teva Pharmaceutical Industries Ltd., Mylan N.V., Bayer AG, Pfizer Inc., GlaxoSmithKline plc, Sanofi S.A., Abbott Laboratories, Merck & Co., Inc., Acelity L.P. Inc., Hisamitsu Pharmaceutical Co., Inc., UCB S.A., HLT, Inc., Zydus Cadila contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Vietnam transdermal drug delivery system market appears promising, driven by increasing healthcare investments and a growing focus on patient-centric solutions. As the healthcare infrastructure expands, more patients will have access to innovative drug delivery methods. Additionally, the integration of digital health technologies is expected to enhance patient monitoring and adherence, further supporting market growth. The emphasis on personalized medicine will also drive demand for tailored transdermal solutions, aligning with global healthcare trends.
| Segment | Sub-Segments |
|---|---|
| By Product | Transdermal Patches Transdermal Gels Transdermal Sprays Others |
| By End-User | Hospitals Ambulatory Surgery Centers Home Care Settings Others |
| By Application | Pain Management Hormonal Therapies Cardiovascular Diseases Central Nervous System Disorders Others |
| By Technology | Passive Systems Active Systems (Iontophoresis, Electroporation) Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Others |
| By Region | Northern Vietnam Central Vietnam Southern Vietnam |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 45 | Product Managers, R&D Directors |
| Healthcare Providers | 80 | Pharmacists, General Practitioners |
| Patients Using Transdermal Systems | 75 | Chronic Pain Patients, Hormone Therapy Patients |
| Regulatory Bodies | 50 | Health Policy Analysts, Regulatory Affairs Specialists |
| Market Research Experts | 60 | Market Analysts, Industry Consultants |
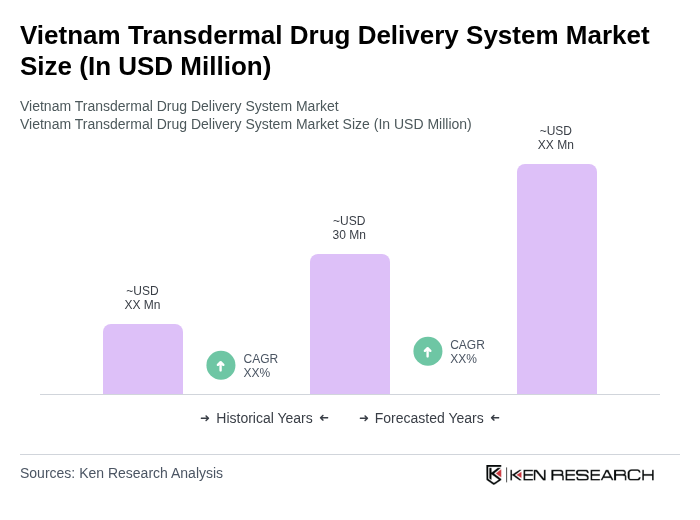
The Vietnam Transdermal Drug Delivery System Market is valued at approximately USD 30 million, driven by the increasing prevalence of chronic diseases and the demand for non-invasive drug delivery methods.