Region:Asia
Author(s):Dev
Product Code:KRAA6127
Pages:81
Published On:January 2026
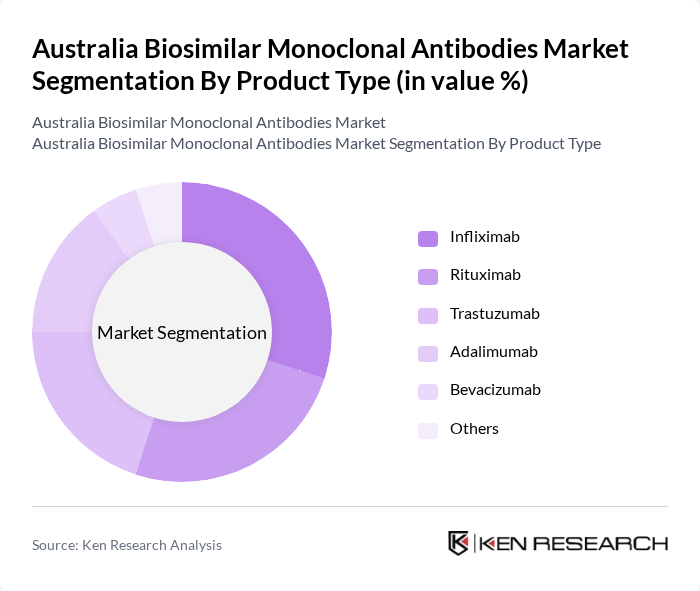
By Product Type:The product type segmentation includes Infliximab, Rituximab, Trastuzumab, Adalimumab, Bevacizumab, and Others. Among these, Infliximab and Rituximab are leading the market due to their widespread use in treating autoimmune diseases and cancers. The increasing acceptance of these biosimilars by healthcare providers and patients is driven by their cost-effectiveness and proven efficacy, making them the preferred choice in many treatment protocols.
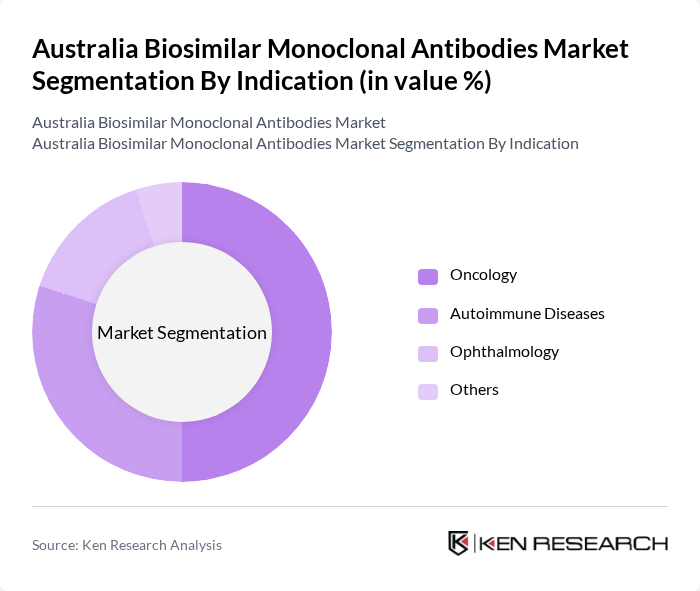
By Indication:The indication segmentation includes Oncology, Autoimmune Diseases, Ophthalmology, and Others. Oncology is the leading segment, driven by the high incidence of cancer and the increasing adoption of biosimilars in cancer treatment protocols. The growing awareness among oncologists regarding the efficacy and safety of biosimilars is contributing to their rising utilization in oncology settings.
The Australia Biosimilar Monoclonal Antibodies Market is characterized by a dynamic mix of regional and international players. Leading participants such as Amgen Inc., Sandoz (Novartis), Pfizer Inc., Mylan N.V., Celltrion Healthcare, Samsung Bioepis, Teva Pharmaceutical Industries Ltd., AbbVie Inc., Boehringer Ingelheim, Roche, Merck & Co., Inc., GSK (GlaxoSmithKline), Hikma Pharmaceuticals, EMD Serono, Fresenius Kabi contribute to innovation, geographic expansion, and service delivery in this space.
The future of the biosimilar monoclonal antibodies market in Australia appears promising, driven by increasing healthcare demands and supportive policies. As chronic diseases continue to rise, the need for affordable treatment options will intensify. Furthermore, advancements in biosimilar development technologies are expected to enhance product quality and efficacy. Collaborations between biosimilar manufacturers and healthcare providers will likely foster greater awareness and acceptance, paving the way for increased market penetration and improved patient outcomes in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Infliximab Rituximab Trastuzumab Adalimumab Bevacizumab Others |
| By Indication | Oncology Autoimmune Diseases Ophthalmology Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Others |
| By Region | New South Wales Victoria Queensland Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Treatment Providers | 100 | Oncologists, Clinical Pharmacists |
| Rheumatology Clinics | 80 | Rheumatologists, Nurse Practitioners |
| Patient Advocacy Groups | 60 | Patient Representatives, Healthcare Advocates |
| Health Insurance Providers | 70 | Policy Analysts, Medical Directors |
| Pharmaceutical Distributors | 90 | Supply Chain Managers, Sales Directors |
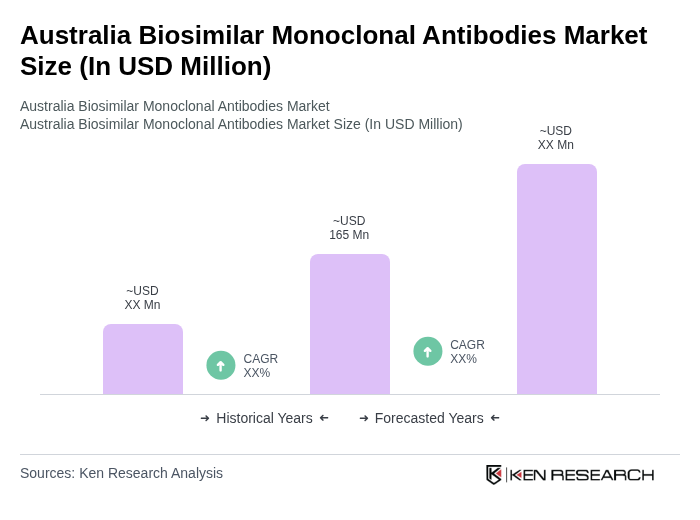
The Australia Biosimilar Monoclonal Antibodies Market is valued at approximately USD 165 million, reflecting a significant growth driven by the increasing prevalence of chronic diseases and the demand for affordable treatment options.