Region:Middle East
Author(s):Dev
Product Code:KRAA6125
Pages:85
Published On:January 2026
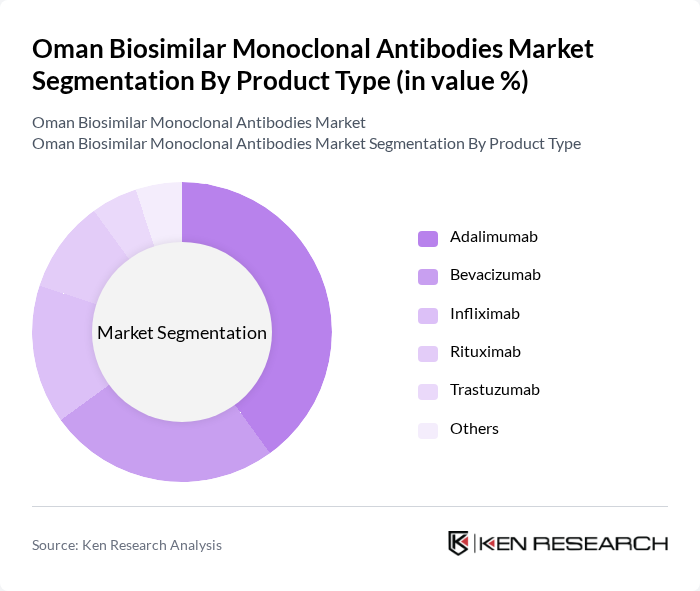
By Product Type:The market is segmented into various product types, including Adalimumab, Bevacizumab, Infliximab, Rituximab, Trastuzumab, and Others. Among these, Adalimumab is the leading product type due to its widespread use in treating autoimmune diseases and its established presence in the market. The increasing demand for effective treatment options for chronic conditions has further solidified its position.
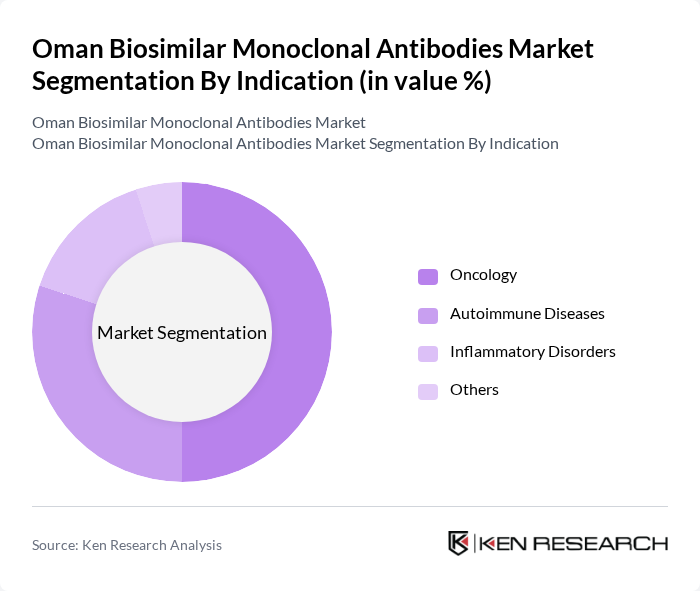
By Indication:The market is categorized based on indications, including Oncology, Autoimmune Diseases, Inflammatory Disorders, and Others. Oncology is the dominant indication, driven by the rising incidence of cancer and the increasing adoption of biosimilars as effective treatment alternatives. The growing focus on personalized medicine and targeted therapies has also contributed to the demand for biosimilars in oncology.
The Oman Biosimilar Monoclonal Antibodies Market is characterized by a dynamic mix of regional and international players. Leading participants such as GlaxoSmithKline plc, Novartis AG, Pfizer Inc., Eli Lilly and Company, Bristol-Myers Squibb, F. Hoffmann-La Roche Ltd., Biogen Inc., Amgen Inc., Sandoz International GmbH, Celltrion Healthcare Co., Ltd., Samsung Bioepis Co., Ltd., Biocon Ltd., Teva Pharmaceutical Industries Ltd., Mylan N.V., AbbVie Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the biosimilar monoclonal antibodies market in Oman appears promising, driven by increasing healthcare investments and a growing awareness of biosimilars among healthcare professionals and patients. As the government continues to enhance healthcare infrastructure, the accessibility of biosimilars is expected to improve. Additionally, the ongoing advancements in biotechnology will likely lead to innovative product developments, further solidifying the role of biosimilars in chronic disease management and personalized medicine.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Adalimumab Bevacizumab Infliximab Rituximab Trastuzumab Others |
| By Indication | Oncology Autoimmune Diseases Inflammatory Disorders Others |
| By End-User | Hospitals Cancer Treatment Centers Specialty Clinics Research Institutions Others |
| By Region | Muscat Dhofar Al Batinah Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Treatment Providers | 100 | Oncologists, Hematologists |
| Pharmaceutical Distributors | 80 | Distribution Managers, Sales Directors |
| Healthcare Policy Makers | 50 | Health Economists, Regulatory Affairs Specialists |
| Patient Advocacy Groups | 70 | Patient Representatives, Care Coordinators |
| Clinical Research Organizations | 60 | Clinical Research Managers, Biostatisticians |
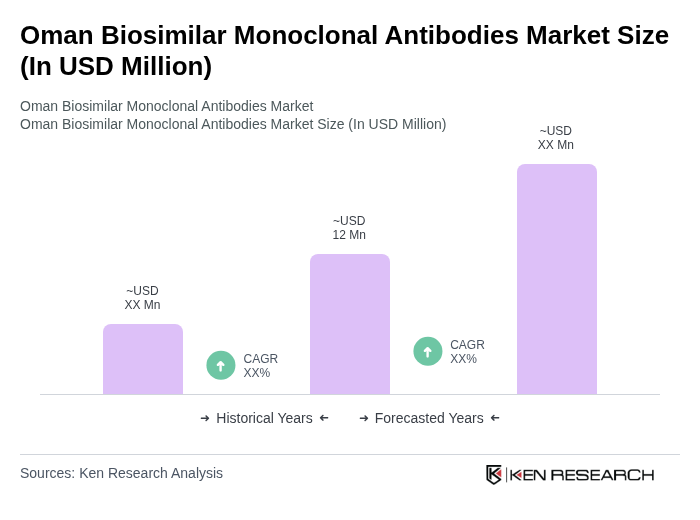
The Oman Biosimilar Monoclonal Antibodies Market is valued at approximately USD 12 million, reflecting a growing demand driven by chronic diseases, healthcare expenditure, and the acceptance of biosimilars among healthcare professionals and patients.