Region:Middle East
Author(s):Dev
Product Code:KRAA6126
Pages:94
Published On:January 2026
By Type:
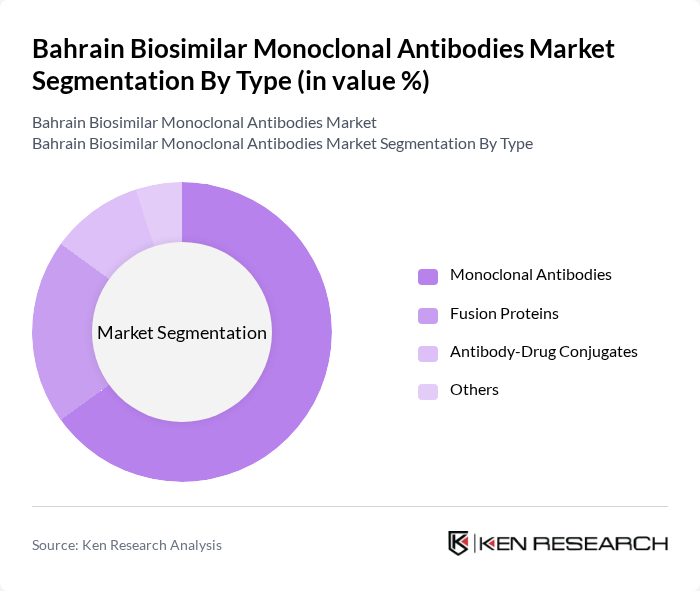
The market is segmented into four main types: Monoclonal Antibodies, Fusion Proteins, Antibody-Drug Conjugates, and Others. Among these, Monoclonal Antibodies dominate the market due to their widespread application in treating various diseases, particularly cancer and autoimmune disorders. The increasing approval of monoclonal biosimilars by regulatory authorities has further fueled their adoption. Fusion Proteins and Antibody-Drug Conjugates are also gaining traction, but they currently hold a smaller market share compared to Monoclonal Antibodies.
By End-User:
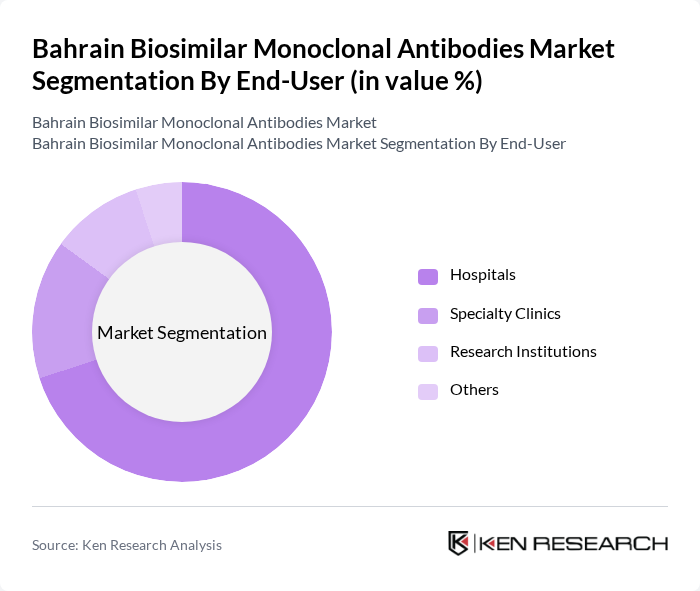
The end-user segmentation includes Hospitals, Specialty Clinics, Research Institutions, and Others. Hospitals are the leading end-users, driven by the high volume of patients requiring treatment for chronic diseases. Specialty Clinics are also significant, particularly for targeted therapies. Research Institutions contribute to the market by developing new biosimilars, while other end-users, such as outpatient facilities, play a smaller role in the overall market.
The Bahrain Biosimilar Monoclonal Antibodies Market is characterized by a dynamic mix of regional and international players. Leading participants such as Amgen Inc., Pfizer Inc., Roche Holding AG, Merck & Co., Inc., Novartis AG, Sanofi S.A., AbbVie Inc., Teva Pharmaceutical Industries Ltd., Celltrion Healthcare Co., Ltd., Mylan N.V., Sandoz International GmbH, Biocon Ltd., GSK (GlaxoSmithKline) plc, Hikma Pharmaceuticals PLC, Fresenius Kabi AG contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Bahrain biosimilar monoclonal antibodies market appears promising, driven by increasing healthcare investments and a growing emphasis on cost-effective treatment options. As the government continues to support biosimilar development through favorable policies and funding, the market is likely to witness enhanced competition and innovation. Additionally, the rising prevalence of chronic diseases will further necessitate the adoption of biosimilars, ensuring that patients have access to affordable and effective therapies in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Fusion Proteins Antibody-Drug Conjugates Others |
| By End-User | Hospitals Specialty Clinics Research Institutions Others |
| By Therapeutic Area | Oncology Autoimmune Diseases Infectious Diseases Others |
| By Distribution Channel | Direct Sales Distributors Online Pharmacies Others |
| By Region | Central Governorate Northern Governorate Southern Governorate Muharraq Governorate |
| By Patient Demographics | Age Group Gender Socioeconomic Status Others |
| By Market Maturity | Emerging Market Established Market Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Treatment Centers | 100 | Oncologists, Clinical Pharmacists |
| Rheumatology Clinics | 80 | Rheumatologists, Nurse Practitioners |
| Pharmaceutical Distributors | 70 | Distribution Managers, Sales Executives |
| Health Insurance Providers | 60 | Policy Analysts, Medical Directors |
| Regulatory Bodies | 50 | Regulatory Affairs Specialists, Compliance Officers |
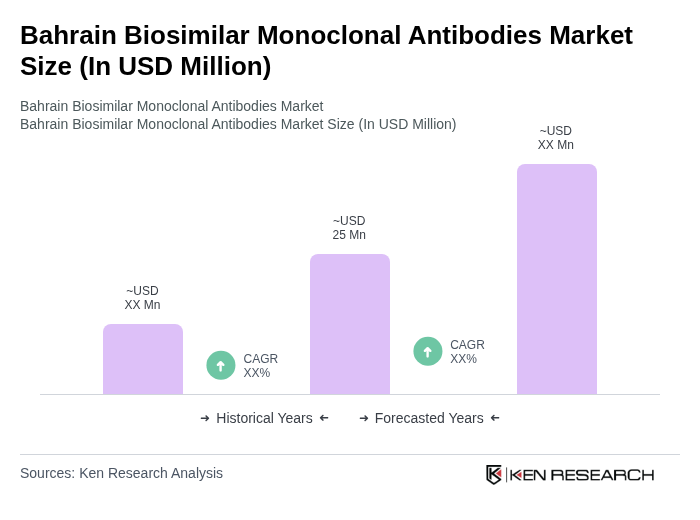
The Bahrain Biosimilar Monoclonal Antibodies Market is valued at approximately USD 25 million, reflecting a growing demand for cost-effective treatment options in response to the increasing prevalence of chronic diseases and rising healthcare costs.