Japan Biosimilar Monoclonal Antibodies Market Overview
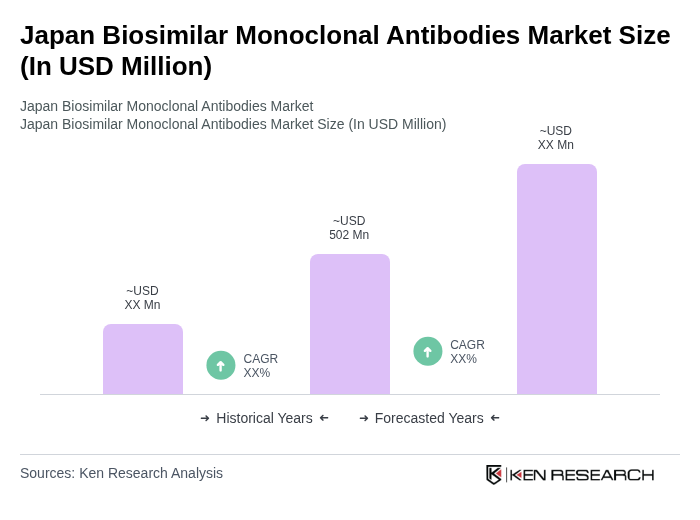
- The Japan Biosimilar Monoclonal Antibodies Market is valued at approximately USD 502 million, based on current market analysis. This growth is primarily driven by the increasing prevalence of chronic diseases, the rising demand for cost-effective treatment options, and the growing acceptance of biosimilars among healthcare professionals and patients. The market is also supported by advancements in biotechnology and regulatory frameworks that facilitate the approval of biosimilars.
- Key regions dominating the market include Tokyo, Osaka, and Nagoya. These cities are home to a high concentration of healthcare facilities, research institutions, and pharmaceutical companies, which contribute to the robust development and distribution of biosimilars. Additionally, the presence of a large patient population and increasing healthcare expenditure in these regions further enhances their market dominance.
- The Japanese government has implemented regulatory frameworks aimed at expediting the approval process for biosimilars. These regulations allow for streamlined review processes, reducing the time required for market entry. The initiative is part of a broader strategy to enhance access to affordable medications and promote competition in the pharmaceutical market, ultimately benefiting patients and healthcare providers.
Japan Biosimilar Monoclonal Antibodies Market Segmentation
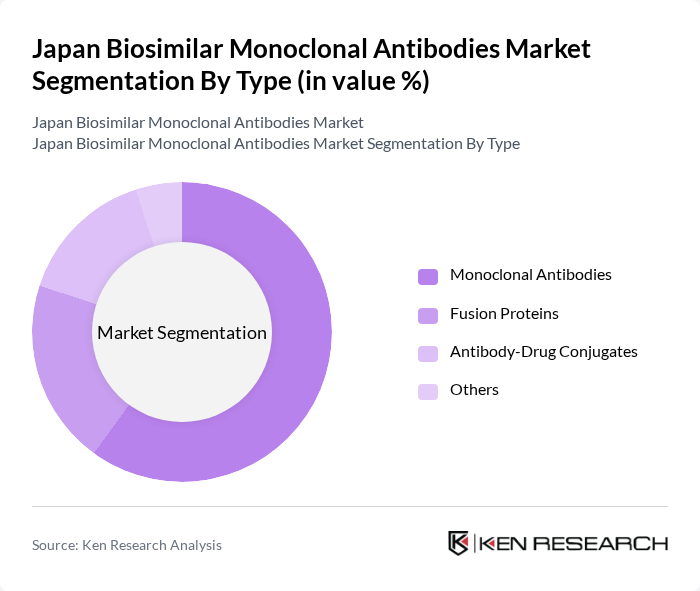
By Type:The market is segmented into various types, including Monoclonal Antibodies, Fusion Proteins, Antibody-Drug Conjugates, and Others. Among these, Monoclonal Antibodies are the leading subsegment due to their widespread application in treating various diseases, particularly in oncology and autoimmune disorders. The increasing adoption of monoclonal antibodies in clinical settings, coupled with their proven efficacy and safety profiles, drives their dominance in the market.
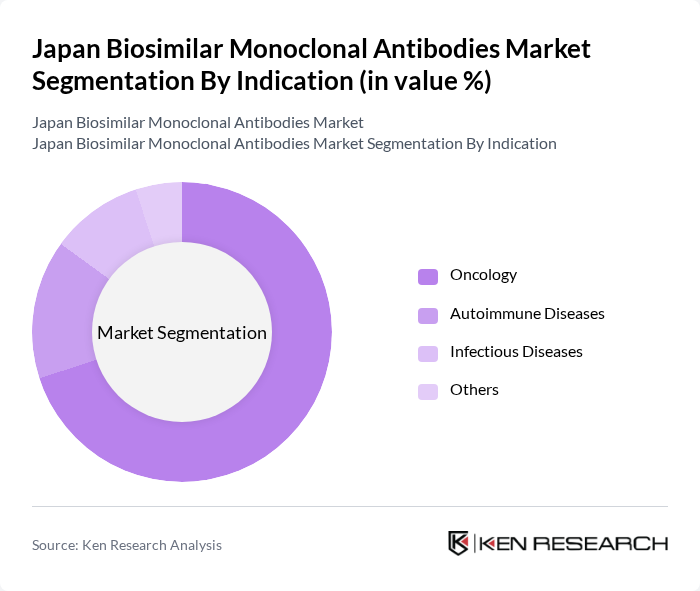
By Indication:The market is categorized based on indications such as Oncology, Autoimmune Diseases, Infectious Diseases, and Others. Oncology is the dominant subsegment, driven by the high incidence of cancer and the increasing use of biosimilars in cancer treatment regimens. The effectiveness of biosimilars in oncology, combined with the growing number of approved biosimilar products, contributes to the significant market share of this segment.
Japan Biosimilar Monoclonal Antibodies Market Competitive Landscape
The Japan Biosimilar Monoclonal Antibodies Market is characterized by a dynamic mix of regional and international players. Leading participants such as Takeda Pharmaceutical Company Limited, Fujifilm Diosynth Biotechnologies, Samsung Bioepis, Amgen Inc., Sandoz International GmbH, Mylan N.V., Biocon Limited, AbbVie Inc., Pfizer Inc., Roche Holding AG, Merck & Co., Inc., Celltrion Healthcare Co., Ltd., EirGenix, Inc., HEC Pharm Co., Ltd., Innovent Biologics, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
Japan Biosimilar Monoclonal Antibodies Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The prevalence of chronic diseases in Japan is rising, with approximately 14 million people living with diabetes. This growing patient population drives demand for effective treatments, including biosimilar monoclonal antibodies. The World Health Organization reported that chronic diseases account for 60% of all deaths in Japan, emphasizing the urgent need for affordable therapeutic options to manage these conditions effectively.
- Rising Healthcare Costs:Japan's healthcare expenditure reached ¥42 trillion (approximately $380 billion), reflecting a 5% increase from the previous year. This surge in costs is prompting healthcare providers and patients to seek more cost-effective treatment alternatives, such as biosimilars. The government is also focusing on reducing overall healthcare spending, which further supports the adoption of biosimilar monoclonal antibodies as a viable solution to manage expenses while maintaining treatment efficacy.
- Expanding Aging Population:In future, Japan's population aged 65 and older is projected to exceed 36 million, representing over 28% of the total population. This demographic shift is associated with an increased incidence of age-related diseases, necessitating innovative treatment options. The demand for biosimilar monoclonal antibodies is expected to rise as healthcare systems adapt to the needs of an aging population, ensuring access to effective therapies for chronic conditions prevalent in older adults.
Market Challenges
- Regulatory Hurdles:The regulatory landscape for biosimilars in Japan is complex, with stringent approval processes that can delay market entry. The Pharmaceuticals and Medical Devices Agency (PMDA) requires extensive clinical data to demonstrate biosimilarity, which can take years to compile. In future, only 10 biosimilars were approved, highlighting the challenges companies face in navigating these regulations while ensuring compliance and safety for patients.
- Competition from Originator Biologics:The presence of established originator biologics poses a significant challenge for biosimilar manufacturers. In future, the market for originator monoclonal antibodies in Japan was valued at ¥1.5 trillion (approximately $13.5 billion), creating a competitive environment. These originator products often have strong brand loyalty and physician preference, making it difficult for biosimilars to gain market share despite their cost advantages.
Japan Biosimilar Monoclonal Antibodies Market Future Outlook
The future of the biosimilar monoclonal antibodies market in Japan appears promising, driven by increasing healthcare demands and a shift towards cost-effective treatment options. As the aging population continues to grow, the need for innovative therapies will intensify. Additionally, advancements in biotechnology and regulatory reforms aimed at expediting biosimilar approvals are expected to enhance market dynamics, fostering a competitive landscape that encourages the development of new biosimilars and improved patient access to essential therapies.
Market Opportunities
- Growing Demand for Cost-Effective Therapies:With healthcare costs rising, there is a significant opportunity for biosimilars to fill the gap in affordable treatment options. The Japanese government is actively promoting the use of biosimilars to reduce healthcare expenditures, which could lead to increased market penetration and acceptance among healthcare providers and patients alike.
- Potential for New Product Launches:The biosimilar market in Japan is ripe for new product introductions, particularly as patents for several originator biologics expire. Companies can capitalize on this opportunity by developing biosimilars that target high-demand therapeutic areas, such as oncology and autoimmune diseases, thereby expanding their product portfolios and enhancing market presence.