Kuwait Biosimilar Monoclonal Antibodies Market Overview
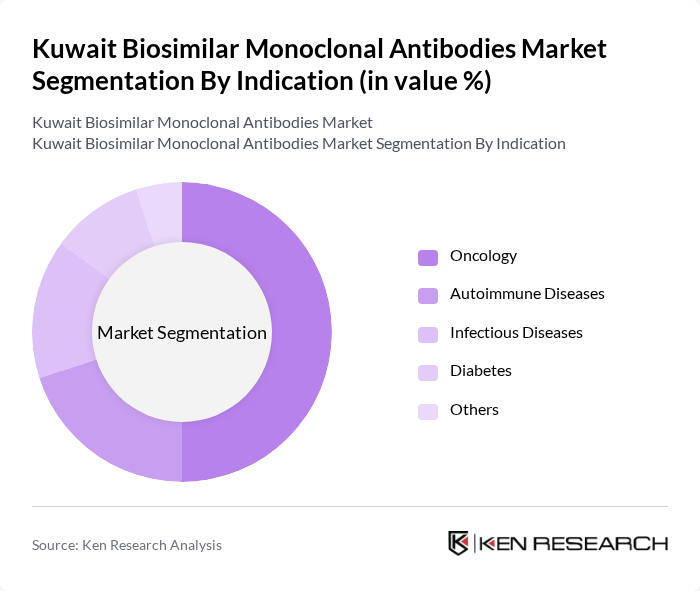
- The Kuwait Biosimilar Monoclonal Antibodies Market is valued at approximately USD 255 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases, rising healthcare costs, and the demand for affordable treatment options. The biosimilars market is gaining traction as healthcare providers and patients seek cost-effective alternatives to expensive biologics, leading to a significant uptick in market activity.
- Kuwait City is the dominant hub for the biosimilar monoclonal antibodies market due to its advanced healthcare infrastructure and a growing population with increasing healthcare needs. The presence of major hospitals and research institutions in the city facilitates the adoption of innovative therapies, while the government's focus on enhancing healthcare services further solidifies its position as a market leader.
- The Kuwaiti regulatory framework supports the approval process for biosimilars through established guidelines for clinical trials and post-marketing surveillance, ensuring that biosimilars meet safety and efficacy standards. This regulatory approach is designed to encourage the entry of new players into the market, fostering competition and ultimately benefiting patients through improved access to affordable therapies.
Kuwait Biosimilar Monoclonal Antibodies Market Segmentation
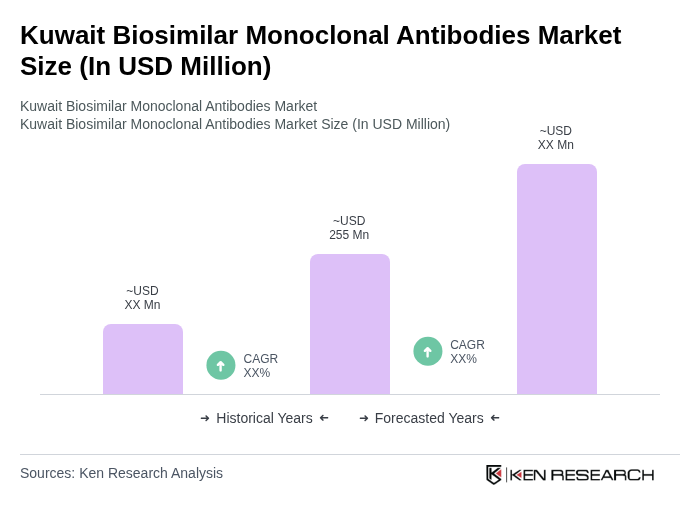
By Molecule Type:The molecule type segmentation includes various categories such as Monoclonal Antibodies, Recombinant Proteins, Insulin, Erythropoietin, Growth Hormones, Interferon, Vaccines, Hormones, Coagulation Factors, and Others. Among these, Monoclonal Antibodies are leading the market due to their widespread application in treating various diseases, particularly in oncology and autoimmune disorders. The increasing acceptance of biosimilars in clinical settings and their cost-effectiveness compared to original biologics are driving their demand. Hormones represent the fastest-growing segment, reflecting broader therapeutic needs and market dynamics.
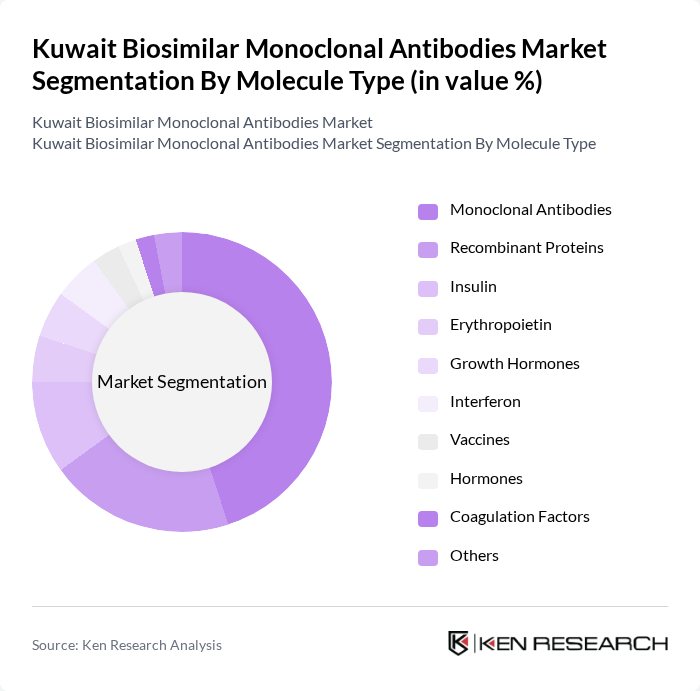
By Indication:The indication segmentation encompasses Oncology, Autoimmune Diseases, Infectious Diseases, Diabetes, and Others. Oncology is the leading segment, driven by the rising incidence of cancer and the increasing adoption of biosimilars in cancer treatment protocols. The effectiveness of monoclonal antibodies in targeting specific cancer cells has made them a preferred choice among healthcare providers, contributing to their dominance in the market.
Kuwait Biosimilar Monoclonal Antibodies Market Competitive Landscape
The Kuwait Biosimilar Monoclonal Antibodies Market is characterized by a dynamic mix of regional and international players. Leading participants such as Amgen Inc., Sandoz International GmbH (Novartis), Pfizer Inc., Viatris Inc. (formerly Mylan N.V.), Teva Pharmaceutical Industries Ltd., Celltrion Healthcare Co., Ltd., Samsung Bioepis Co., Ltd., AbbVie Inc., Biocon Ltd., GlaxoSmithKline plc (GSK), Merck & Co., Inc., Novartis AG, Roche Holding AG, Eli Lilly and Company, and Sanofi S.A. contribute to innovation, geographic expansion, and service delivery in this space.
Kuwait Biosimilar Monoclonal Antibodies Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The rise in chronic diseases such as diabetes and cancer in Kuwait is a significant growth driver for the biosimilar monoclonal antibodies market. According to the World Health Organization, approximately 32% of the Kuwaiti population suffers from chronic conditions, leading to a higher demand for effective treatment options. This growing patient base is expected to increase the utilization of biosimilars, which offer similar efficacy at lower costs, thereby enhancing patient access to necessary therapies.
- Rising Healthcare Expenditure:Kuwait's healthcare expenditure is projected to reach approximately $11 billion in future, reflecting a commitment to improving healthcare services. This increase in spending is driven by government initiatives aimed at enhancing healthcare infrastructure and services. As healthcare budgets expand, there is a greater opportunity for the adoption of biosimilars, which can provide cost-effective alternatives to expensive originator biologics, thus alleviating financial pressures on the healthcare system.
- Demand for Cost-Effective Treatment Options:The financial burden of healthcare in Kuwait has prompted a shift towards more cost-effective treatment options, including biosimilars. With the average annual cost of biologic therapies exceeding $32,000, healthcare providers are increasingly looking for alternatives that maintain therapeutic efficacy while reducing costs. The growing acceptance of biosimilars among healthcare professionals and patients is expected to drive market growth, as these products can significantly lower treatment expenses without compromising quality.
Market Challenges
- Regulatory Hurdles:The regulatory landscape for biosimilars in Kuwait presents significant challenges, including lengthy approval processes and stringent requirements for clinical data. The Kuwait Food and Drug Administration (KFDA) has established rigorous guidelines that can delay market entry for biosimilars. In future, it is estimated that the average time for biosimilar approval could exceed 20 months, hindering timely access to these essential therapies for patients in need.
- Limited Awareness Among Healthcare Professionals:Despite the potential benefits of biosimilars, there remains a notable lack of awareness and understanding among healthcare professionals in Kuwait. A survey conducted by the Kuwait Medical Association indicated that over 45% of physicians are unfamiliar with biosimilar products and their therapeutic equivalence to originator biologics. This knowledge gap can lead to hesitancy in prescribing biosimilars, ultimately limiting their market penetration and growth potential.
Kuwait Biosimilar Monoclonal Antibodies Market Future Outlook
The future of the biosimilar monoclonal antibodies market in Kuwait appears promising, driven by increasing healthcare investments and a growing emphasis on cost-effective treatment solutions. As the government continues to enhance healthcare infrastructure, the accessibility of biosimilars is expected to improve. Additionally, ongoing collaborations between local manufacturers and international firms will likely foster innovation and expedite the development of new biosimilar products, further solidifying their role in the healthcare landscape.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Kuwaiti government is investing heavily in healthcare infrastructure, with plans to allocate $2 billion for new hospitals and clinics in future. This expansion will create a conducive environment for the introduction and distribution of biosimilars, enhancing patient access to these therapies and driving market growth.
- Collaborations with Local Manufacturers:Strategic partnerships between international biosimilar developers and local manufacturers can significantly enhance production capabilities. By leveraging local expertise and resources, these collaborations can reduce costs and improve supply chain efficiencies, making biosimilars more accessible to the Kuwaiti market and increasing their adoption among healthcare providers.