About the Report
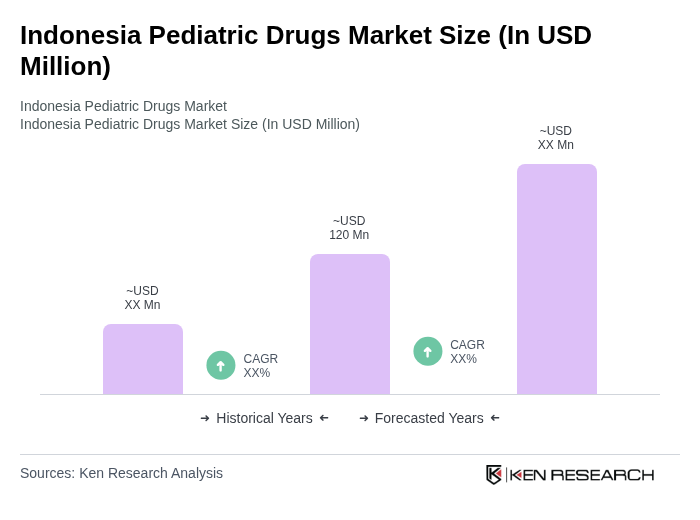
Base Year 2024Indonesia Pediatric Drugs Market Overview
- The Indonesia Pediatric Drugs Market is valued at USD 120 million, based on a five-year historical analysis. This growth is primarily driven by increasing healthcare expenditure, rising awareness of pediatric health issues, a growing population of children requiring medical attention, greater knowledge of home healthcare, technological advancements, and rising disposable incomes. The market has seen a surge in demand for specialized pediatric medications, particularly in urban areas where healthcare access is improving.
- Key cities such as Jakarta, Surabaya, and Bandung dominate the market due to their large populations and advanced healthcare infrastructure. These urban centers have a higher concentration of hospitals and clinics, leading to increased availability and accessibility of pediatric drugs. Additionally, the presence of major pharmaceutical companies in these cities contributes to their market dominance.
- The Minister of Health Regulation No. 2 of 2024 issued by the Ministry of Health requires pediatric drugs to meet specific efficacy, safety, and quality standards tailored for children, including appropriate dosage forms, clinical data from pediatric populations, and stability testing under child-use conditions. Manufacturers must submit child-specific pharmacokinetic and pharmacodynamic studies for registration, ensuring compliance through BPOM approval processes with defined thresholds for excipients and labeling requirements.
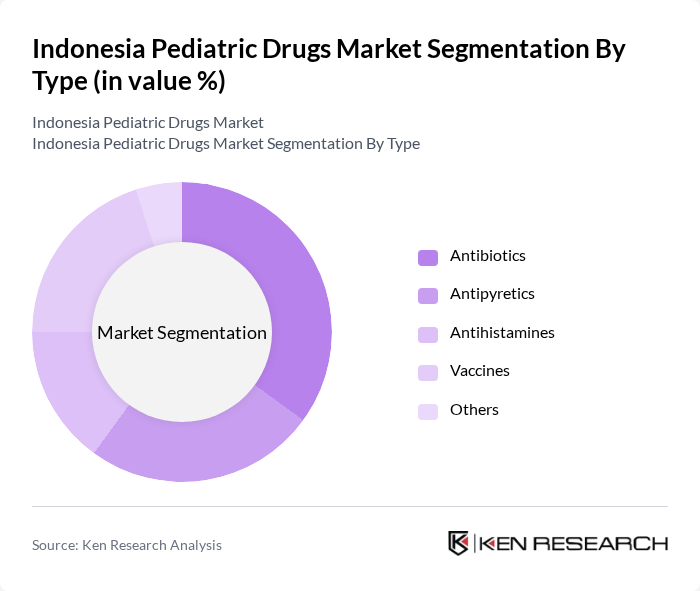
Indonesia Pediatric Drugs Market Segmentation
By Type:The market is segmented into various types of pediatric drugs, including antibiotics, antipyretics, antihistamines, vaccines, and others. Among these, antibiotics and vaccines are particularly significant due to the high prevalence of infectious diseases in children and the ongoing vaccination programs. The demand for antipyretics also remains strong, driven by the common occurrence of fevers in pediatric patients. The increasing focus on preventive healthcare is further boosting the vaccine segment.
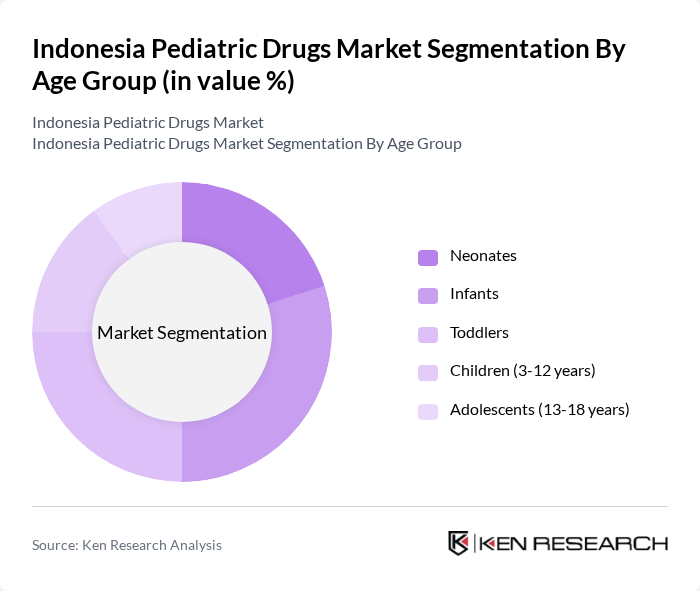
By Age Group:The pediatric drugs market is also segmented by age groups, including neonates, infants, toddlers, children (3-12 years), and adolescents (13-18 years). The infant segment is particularly dominant due to the high incidence of health issues requiring medication in this age group. Additionally, the growing awareness among parents regarding child health and the importance of timely medical intervention contribute to the increasing demand for pediatric drugs across all age groups.
Indonesia Pediatric Drugs Market Competitive Landscape
The Indonesia Pediatric Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT Kalbe Farma Tbk, PT Kimia Farma Tbk, PT Indofarma Tbk, PT Merck Tbk, PT Sanofi Indonesia, PT Pfizer Indonesia, PT Novartis Indonesia, PT AstraZeneca Indonesia, PT GlaxoSmithKline Indonesia, PT Bayer Indonesia, PT Abbott Indonesia, PT Johnson & Johnson Indonesia, PT Roche Indonesia, PT Sandoz Indonesia, PT Darya-Varia Laboratoria Tbk contribute to innovation, geographic expansion, and service delivery in this space.
Indonesia Pediatric Drugs Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Pediatric Diseases:The rise in pediatric diseases in Indonesia is significant, with the World Health Organization reporting that respiratory infections affect approximately 1.5 million children annually. Additionally, the prevalence of childhood obesity has increased by 30% over the last decade, leading to a surge in demand for pediatric medications. This growing health burden necessitates a robust supply of effective pediatric drugs, driving market growth and innovation in treatment options tailored for children.
- Rising Healthcare Expenditure:Indonesia's healthcare expenditure is projected to reach IDR 1,500 trillion (approximately USD 100 billion) in future, reflecting a 10% increase from previous levels. This rise is fueled by government initiatives aimed at improving healthcare access and quality. Increased funding allows for better procurement of pediatric drugs and enhances healthcare facilities, ultimately improving treatment outcomes for children and expanding the pediatric drugs market significantly.
- Growing Awareness of Pediatric Health:Awareness campaigns and educational programs have led to a 25% increase in pediatric health consultations over the past five years. The Indonesian government, alongside NGOs, has invested heavily in public health initiatives, resulting in improved health literacy among parents. This heightened awareness drives demand for pediatric drugs, as parents are more proactive in seeking medical care and treatment options for their children, thus expanding the market.
Market Challenges
- Limited Access to Healthcare in Rural Areas:Approximately 60% of Indonesia's population resides in rural areas, where healthcare access remains a significant challenge. The World Bank estimates that only 30% of rural health facilities are adequately equipped to provide pediatric care. This disparity limits the availability of pediatric drugs and treatment options, hindering market growth and leaving many children without necessary medical attention.
- Regulatory Hurdles:The Indonesian pharmaceutical sector faces stringent regulatory requirements, which can delay the approval of pediatric drugs. The National Agency of Drug and Food Control (BPOM) has reported that the average approval time for new pediatric formulations can exceed 18 months. These regulatory challenges can deter investment in pediatric drug development and limit the availability of innovative treatments in the market.
Indonesia Pediatric Drugs Market Future Outlook
The future of the pediatric drugs market in Indonesia appears promising, driven by increasing healthcare investments and a focus on improving pediatric health outcomes. As the government continues to enhance healthcare infrastructure and access, the demand for pediatric medications is expected to rise. Additionally, advancements in telemedicine and digital health solutions will facilitate better healthcare delivery, ensuring that children receive timely and effective treatments, thereby fostering market growth in the coming years.
Market Opportunities
- Development of Generic Pediatric Drugs:The growing demand for affordable healthcare solutions presents a significant opportunity for the development of generic pediatric drugs. With the Indonesian government encouraging local manufacturing, companies can capitalize on this trend to provide cost-effective alternatives, improving access to essential medications for children across the country.
- Collaborations with NGOs for Healthcare Access:Partnerships with non-governmental organizations can enhance healthcare access in underserved areas. By collaborating on outreach programs and health education initiatives, pharmaceutical companies can improve the distribution of pediatric drugs, ensuring that vulnerable populations receive necessary treatments and fostering brand loyalty in the process.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Antibiotics Antipyretics Antihistamines Vaccines Others |
| By Age Group | Neonates Infants Toddlers Children (3-12 years) Adolescents (13-18 years) |
| By Distribution Channel | Hospital pharmacies Retail pharmacies Online pharmacies Direct sales Others |
| By Therapeutic Area | Infectious diseases Respiratory disorders Gastrointestinal disorders Neurological disorders Others |
| By Formulation Type | Liquid formulations Solid formulations Semi-solid formulations Others |
| By Route of Administration | Oral Injectable Topical Others |
| By End-User | Hospitals Clinics Home care Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Badan Pengawas Obat dan Makanan - BPOM)
Manufacturers and Producers
Distributors and Retailers
Pharmaceutical Wholesalers
Healthcare Providers and Pediatric Clinics
Industry Associations (e.g., Indonesian Pharmaceutical Association)
Health Insurance Companies
Players Mentioned in the Report:
PT Kalbe Farma Tbk
PT Kimia Farma Tbk
PT Indofarma Tbk
PT Merck Tbk
PT Sanofi Indonesia
PT Pfizer Indonesia
PT Novartis Indonesia
PT AstraZeneca Indonesia
PT GlaxoSmithKline Indonesia
PT Bayer Indonesia
PT Abbott Indonesia
PT Johnson & Johnson Indonesia
PT Roche Indonesia
PT Sandoz Indonesia
PT Darya-Varia Laboratoria Tbk
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia Pediatric Drugs Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia Pediatric Drugs Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia Pediatric Drugs Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of pediatric diseases
3.1.2 Rising healthcare expenditure
3.1.3 Growing awareness of pediatric health
3.1.4 Expansion of healthcare infrastructure
3.2 Market Challenges
3.2.1 Limited access to healthcare in rural areas
3.2.2 Regulatory hurdles
3.2.3 High cost of pediatric drugs
3.2.4 Lack of pediatric-specific formulations
3.3 Market Opportunities
3.3.1 Development of generic pediatric drugs
3.3.2 Increasing investment in pediatric research
3.3.3 Collaborations with NGOs for healthcare access
3.3.4 Digital health solutions for pediatric care
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Growth of telemedicine in pediatric care
3.4.3 Increasing focus on preventive healthcare
3.4.4 Rise in demand for over-the-counter pediatric medications
3.5 Government Regulation
3.5.1 Stricter regulations on drug safety
3.5.2 Policies promoting pediatric drug research
3.5.3 Incentives for local drug manufacturers
3.5.4 Guidelines for pediatric clinical trials
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia Pediatric Drugs Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia Pediatric Drugs Market Segmentation
8.1 By Type
8.1.1 Antibiotics
8.1.2 Antipyretics
8.1.3 Antihistamines
8.1.4 Vaccines
8.1.5 Others
8.2 By Age Group
8.2.1 Neonates
8.2.2 Infants
8.2.3 Toddlers
8.2.4 Children (3-12 years)
8.2.5 Adolescents (13-18 years)
8.3 By Distribution Channel
8.3.1 Hospital pharmacies
8.3.2 Retail pharmacies
8.3.3 Online pharmacies
8.3.4 Direct sales
8.3.5 Others
8.4 By Therapeutic Area
8.4.1 Infectious diseases
8.4.2 Respiratory disorders
8.4.3 Gastrointestinal disorders
8.4.4 Neurological disorders
8.4.5 Others
8.5 By Formulation Type
8.5.1 Liquid formulations
8.5.2 Solid formulations
8.5.3 Semi-solid formulations
8.5.4 Others
8.6 By Route of Administration
8.6.1 Oral
8.6.2 Injectable
8.6.3 Topical
8.6.4 Others
8.7 By End-User
8.7.1 Hospitals
8.7.2 Clinics
8.7.3 Home care
8.7.4 Others
9. Indonesia Pediatric Drugs Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Diversification Index
9.2.8 R&D Investment as a Percentage of Revenue
9.2.9 Distribution Network Efficiency
9.2.10 Brand Recognition Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 PT Kalbe Farma Tbk
9.5.2 PT Kimia Farma Tbk
9.5.3 PT Indofarma Tbk
9.5.4 PT Merck Tbk
9.5.5 PT Sanofi Indonesia
9.5.6 PT Pfizer Indonesia
9.5.7 PT Novartis Indonesia
9.5.8 PT AstraZeneca Indonesia
9.5.9 PT GlaxoSmithKline Indonesia
9.5.10 PT Bayer Indonesia
9.5.11 PT Abbott Indonesia
9.5.12 PT Johnson & Johnson Indonesia
9.5.13 PT Roche Indonesia
9.5.14 PT Sandoz Indonesia
9.5.15 PT Darya-Varia Laboratoria Tbk
10. Indonesia Pediatric Drugs Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Social Affairs
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Hospital infrastructure
10.2.2 Pharmaceutical supply chains
10.2.3 Healthcare technology
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Home care providers
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Healthcare professionals
10.4.2 Parents and guardians
10.4.3 Pediatric patients
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost savings
10.5.2 Improved health outcomes
10.5.3 Increased patient satisfaction
10.5.4 Others
11. Indonesia Pediatric Drugs Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban retail vs rural NGO tie-ups
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands
5. Unmet Demand & Latent Needs
5.1 Category gaps
5.2 Consumer segments
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
7. Value Proposition
7.1 Sustainability
7.2 Integrated supply chains
8. Key Activities
8.1 Regulatory compliance
8.2 Branding
8.3 Distribution setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix
9.1.2 Pricing band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target countries
9.2.2 Compliance roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of government health reports and pediatric drug regulations in Indonesia
- Review of market reports from pharmaceutical associations and healthcare organizations
- Examination of published studies on pediatric health trends and drug utilization
Primary Research
- Interviews with pediatricians and healthcare professionals across major hospitals
- Surveys with pharmacists regarding pediatric drug prescriptions and sales
- Focus groups with parents to understand perceptions and preferences for pediatric medications
Validation & Triangulation
- Cross-validation of findings with multiple healthcare stakeholders
- Triangulation of data from government health statistics and industry reports
- Sanity checks through expert panel reviews comprising pediatric specialists
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total healthcare expenditure in Indonesia allocated to pediatric care
- Analysis of demographic data to determine the pediatric population size and growth
- Incorporation of national health initiatives aimed at improving child health outcomes
Bottom-up Modeling
- Volume estimates based on prescription data from hospitals and clinics
- Cost analysis of pediatric drugs based on pricing from major pharmaceutical distributors
- Calculation of market size using volume x price methodology for various drug categories
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating economic growth and healthcare access
- Scenario modeling based on potential changes in healthcare policy and drug pricing
- Baseline, optimistic, and pessimistic projections for pediatric drug market growth through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pediatricians in Urban Hospitals | 120 | Pediatric Specialists, General Practitioners |
| Pharmacists in Community Pharmacies | 100 | Pharmacy Managers, Retail Pharmacists |
| Parents of Pediatric Patients | 120 | Parents, Guardians |
| Healthcare Administrators | 80 | Hospital Administrators, Health Policy Makers |
| Medical Researchers in Pediatrics | 70 | Clinical Researchers, Academic Professionals |
Frequently Asked Questions
What is the current value of the Indonesia Pediatric Drugs Market?
The Indonesia Pediatric Drugs Market is valued at approximately USD 120 million, reflecting a significant growth driven by increased healthcare expenditure, rising awareness of pediatric health issues, and a growing population of children requiring medical attention.