Region:Asia
Author(s):Rebecca
Product Code:KRAA0677
Pages:89
Published On:January 2026
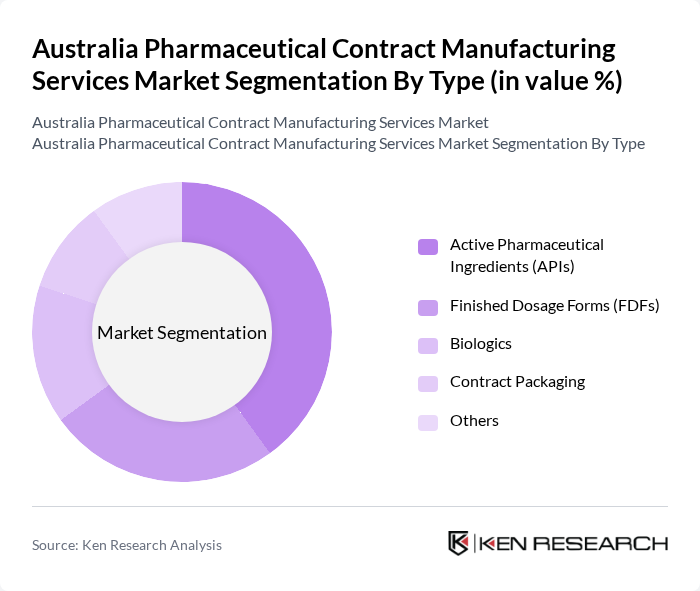
By Type:The market is segmented into various types, including Active Pharmaceutical Ingredients (APIs), Finished Dosage Forms (FDFs), Biologics, Contract Packaging, and Others. Among these, the Active Pharmaceutical Ingredients (APIs) segment is currently leading the market due to the increasing demand for generic drugs and the growing trend of outsourcing API production to reduce costs and enhance efficiency.
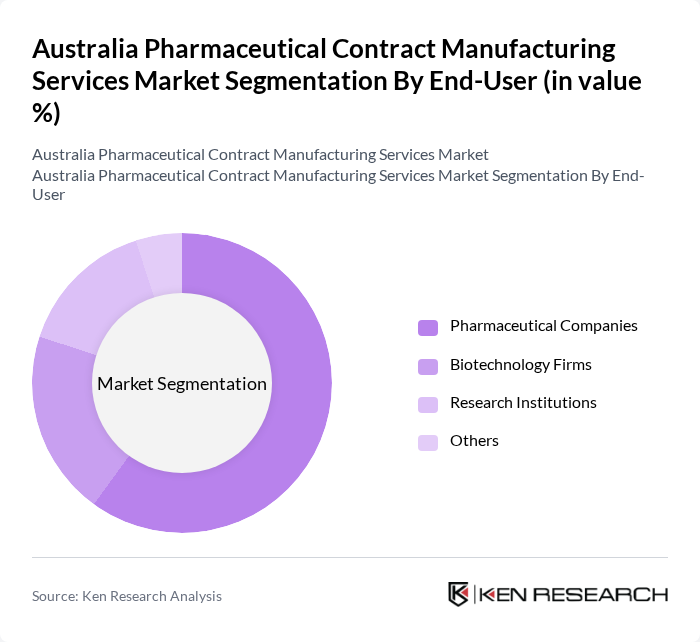
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Research Institutions, and Others. Pharmaceutical Companies dominate this segment as they increasingly rely on contract manufacturing services to enhance their production capabilities and focus on research and development, thus driving the demand for these services.
The Australia Pharmaceutical Contract Manufacturing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as CSL Limited, Mayne Pharma Group Limited, IDT Australia Limited, Sigma Healthcare Limited, Patheon (Thermo Fisher Scientific), Lonza Group AG, Catalent, Inc., Recipharm AB, Fareva, Aenova Group, Piramal Pharma Solutions, Almac Group, Famar, Bausch Health Companies Inc., Sandoz (Novartis) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Australian pharmaceutical contract manufacturing services market appears promising, driven by advancements in technology and an increasing focus on personalized medicine. As companies adopt automation and digital transformation strategies, operational efficiencies are expected to improve significantly. Additionally, the growing trend towards personalized medicine will create new opportunities for contract manufacturers to develop tailored therapies, enhancing their relevance in a rapidly evolving healthcare landscape.
| Segment | Sub-Segments |
|---|---|
| By Type | Active Pharmaceutical Ingredients (APIs) Finished Dosage Forms (FDFs) Biologics Contract Packaging Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Research Institutions Others |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Others |
| By Region | New South Wales Victoria Queensland Western Australia Others |
| By Manufacturing Process | Batch Manufacturing Continuous Manufacturing Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Service Type | Contract Manufacturing Contract Research Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Contract Manufacturing | 150 | Manufacturing Directors, Operations Managers |
| Regulatory Compliance in Pharma | 100 | Quality Assurance Managers, Regulatory Affairs Specialists |
| Biopharmaceutical Manufacturing Services | 80 | Biotech Executives, Production Supervisors |
| Packaging and Labeling Services | 70 | Packaging Engineers, Supply Chain Coordinators |
| Contract Research Organizations (CROs) | 90 | Clinical Operations Managers, Project Leads |
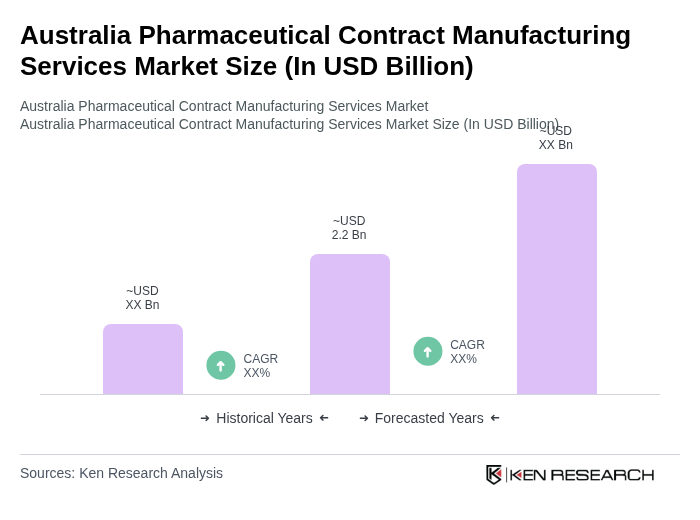
The Australia Pharmaceutical Contract Manufacturing Services Market is valued at approximately USD 2.2 billion, reflecting a significant growth trend driven by the increasing demand for outsourcing manufacturing processes by pharmaceutical companies.