Region:Europe
Author(s):Dev
Product Code:KRAA4904
Pages:86
Published On:September 2025
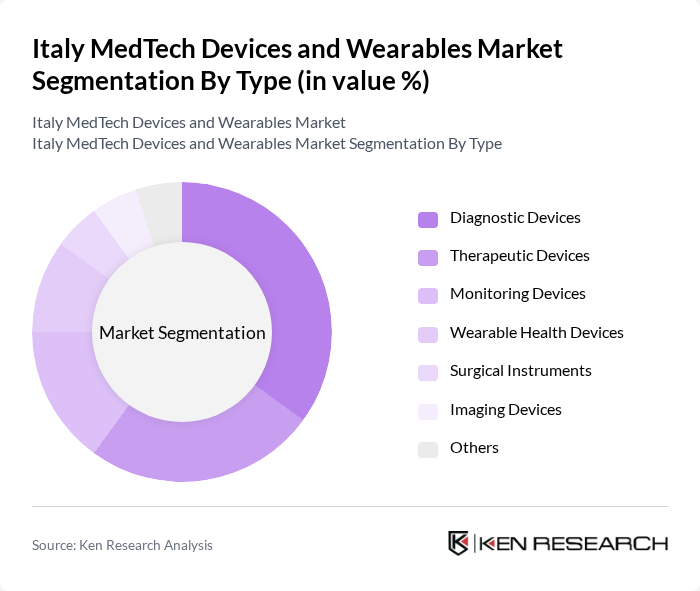
By Type:The market is segmented into various types, including Diagnostic Devices, Therapeutic Devices, Monitoring Devices, Wearable Health Devices, Surgical Instruments, Imaging Devices, and Others. Among these, Diagnostic Devices are currently leading the market due to the increasing demand for early disease detection and the growing adoption of point-of-care testing. The rise in chronic diseases and the need for timely diagnosis have made this segment particularly significant.
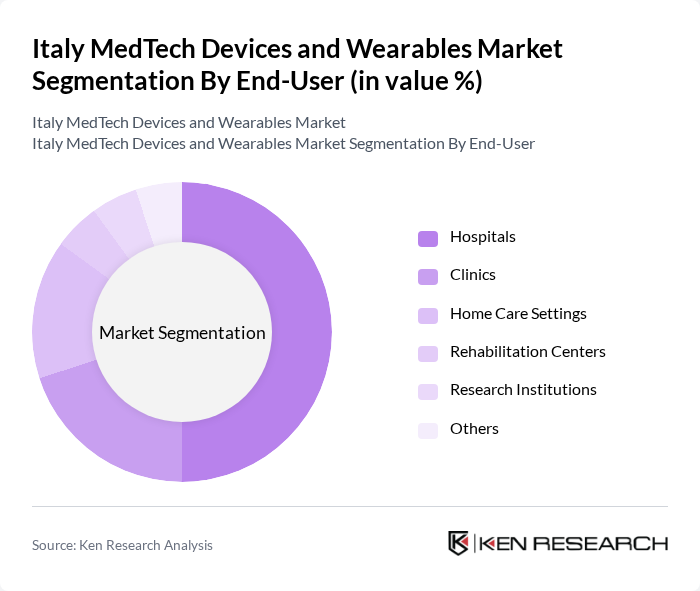
By End-User:The end-user segmentation includes Hospitals, Clinics, Home Care Settings, Rehabilitation Centers, Research Institutions, and Others. Hospitals are the dominant end-user segment, driven by the increasing number of surgical procedures and the demand for advanced medical technologies. The growing focus on patient care and the need for efficient healthcare delivery systems have further solidified hospitals' position in the market.
The Italy MedTech Devices and Wearables Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Siemens Healthineers, Philips Healthcare, GE Healthcare, Abbott Laboratories, Boston Scientific, Johnson & Johnson, Stryker Corporation, B. Braun Melsungen AG, Zimmer Biomet, Roche Diagnostics, Canon Medical Systems, Olympus Corporation, Mindray Medical International Limited, Terumo Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Italy MedTech devices and wearables market appears promising, driven by technological innovations and an increasing focus on preventive healthcare. As the healthcare landscape evolves, the integration of AI and machine learning into devices will enhance patient monitoring and treatment personalization. Furthermore, the expansion of telehealth services will facilitate greater access to healthcare, particularly in rural areas. These trends indicate a robust growth trajectory, with significant opportunities for companies that can navigate regulatory challenges and address consumer needs effectively.
| Segment | Sub-Segments |
|---|---|
| By Type | Diagnostic Devices Therapeutic Devices Monitoring Devices Wearable Health Devices Surgical Instruments Imaging Devices Others |
| By End-User | Hospitals Clinics Home Care Settings Rehabilitation Centers Research Institutions Others |
| By Application | Cardiovascular Diabetes Management Neurology Orthopedics Respiratory Others |
| By Distribution Channel | Direct Sales Online Sales Retail Pharmacies Medical Supply Stores Others |
| By Price Range | Low-End Devices Mid-Range Devices High-End Devices |
| By Technology | Wearable Sensors Mobile Health Applications Cloud Computing Solutions Telemedicine Platforms |
| By Regulatory Compliance | CE Marked Products FDA Approved Products ISO Certified Products Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers Using Wearables | 150 | Doctors, Nurses, Health Technologists |
| Patients Utilizing MedTech Devices | 120 | Chronic Disease Patients, Elderly Users |
| Manufacturers of MedTech Devices | 100 | Product Managers, R&D Directors |
| Regulatory Bodies and Health Authorities | 80 | Regulatory Affairs Specialists, Policy Makers |
| Technology Developers in Wearable Sector | 90 | Software Engineers, Product Developers |
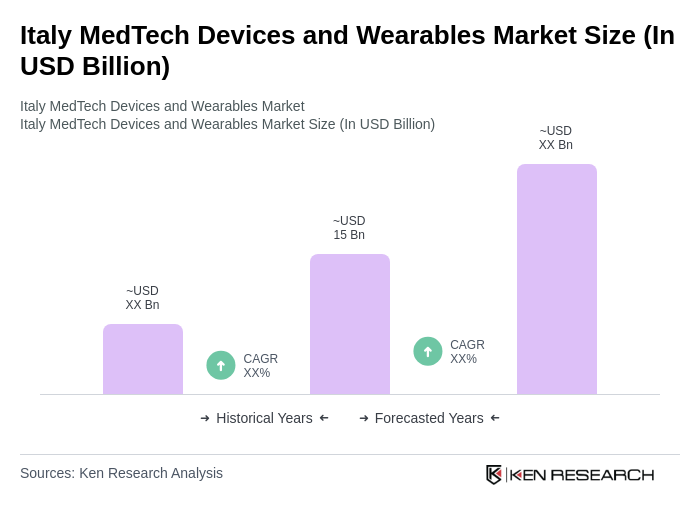
The Italy MedTech Devices and Wearables Market is valued at approximately USD 15 billion, reflecting significant growth driven by technological advancements, increased healthcare spending, and a rising demand for personalized healthcare solutions.