Region:Middle East
Author(s):Rebecca
Product Code:KRAB2052
Pages:87
Published On:January 2026
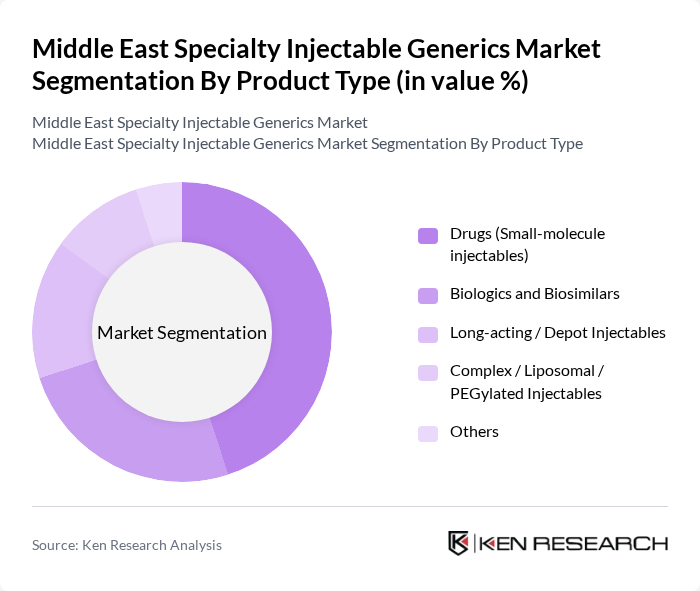
By Product Type:The product type segmentation includes various categories such as Drugs (Small-molecule injectables), Biologics and Biosimilars, Long-acting / Depot Injectables, Complex / Liposomal / PEGylated Injectables, and Others. Among these, Drugs (Small-molecule injectables) are leading the market due to their widespread use in treating various conditions, including infections and chronic diseases, and their relatively lower development and manufacturing costs compared with complex biologics. The increasing demand for affordable treatment options, strong government support for generics and off?patent small molecules, and the growing number of approved small-molecule injectables in therapeutic areas such as oncology, anti?infectives, and cardiovascular diseases are driving this segment's growth.
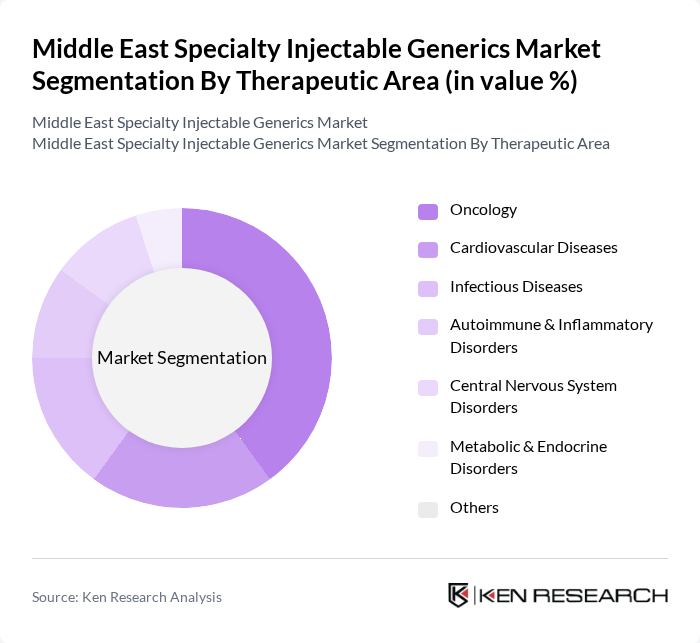
By Therapeutic Area:The therapeutic area segmentation encompasses Oncology, Cardiovascular Diseases, Infectious Diseases, Autoimmune & Inflammatory Disorders, Central Nervous System Disorders, Metabolic & Endocrine Disorders, and Others. Oncology is the leading therapeutic area, driven by the rising incidence of cancer in the Middle East, the high cost of branded oncology biologics, and the increasing availability of effective injectable generic and biosimilar cancer therapies. The growing focus on personalized medicine and targeted parenteral therapies, alongside expanding use of monoclonal antibodies and other high?value injectables in oncology, is also contributing to the expansion of this segment, with similar trends observed for specialty generics in cardiovascular and autoimmune diseases.
The Middle East Specialty Injectable Generics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Hikma Pharmaceuticals PLC, Julphar (Gulf Pharmaceutical Industries), SPIMACO (Saudi Pharmaceutical Industries & Medical Appliances Corporation), Tabuk Pharmaceuticals, Jamjoom Pharma, Eva Pharma, Pharma International Company (PIC), Dar Al Dawa Development & Investment Co., Arab Pharmaceutical Manufacturing Co., AJA Pharma, Pfizer Inc., Teva Pharmaceutical Industries Ltd., Sandoz (Novartis AG), Viatris Inc. (formerly Mylan N.V.), Fresenius Kabi AG contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Middle East specialty injectable generics market appears promising, driven by increasing healthcare investments and a growing focus on chronic disease management. As healthcare systems evolve, the integration of digital health solutions and telemedicine is expected to enhance patient access to injectable therapies. Furthermore, the ongoing collaboration between local manufacturers and international firms will likely foster innovation, leading to the development of new formulations that cater to the region's unique healthcare needs.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Drugs (Small-molecule injectables) Biologics and Biosimilars Long-acting / Depot Injectables Complex / Liposomal / PEGylated Injectables Others |
| By Therapeutic Area | Oncology Cardiovascular Diseases Infectious Diseases Autoimmune & Inflammatory Disorders Central Nervous System Disorders Metabolic & Endocrine Disorders Others |
| By End-User | Public Hospitals Private Hospitals Specialty Clinics & Infusion Centers Retail & Community Pharmacies Homecare & Ambulatory Settings Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Specialty Pharmacies Government Procurement & Tenders Wholesalers / Distributors Online Pharmacies Others |
| By Packaging / Container Type | Vials Ampoules Pre-filled Syringes Cartridges & Pens Premix & Large-volume Parenterals Others |
| By Route of Administration | Intravenous (IV) Intramuscular (IM) Subcutaneous (SC) Intrathecal & Epidural Others |
| By Country / Sub-region | Saudi Arabia United Arab Emirates Qatar Kuwait Oman Bahrain Egypt Jordan & Levant Rest of Middle East & North Africa |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Procurement Departments | 120 | Procurement Managers, Supply Chain Directors |
| Pharmacy Chains | 90 | Pharmacy Managers, Inventory Specialists |
| Healthcare Providers | 75 | Doctors, Nurse Practitioners |
| Regulatory Bodies | 45 | Regulatory Affairs Managers, Compliance Officers |
| Market Research Analysts | 60 | Market Analysts, Business Development Managers |
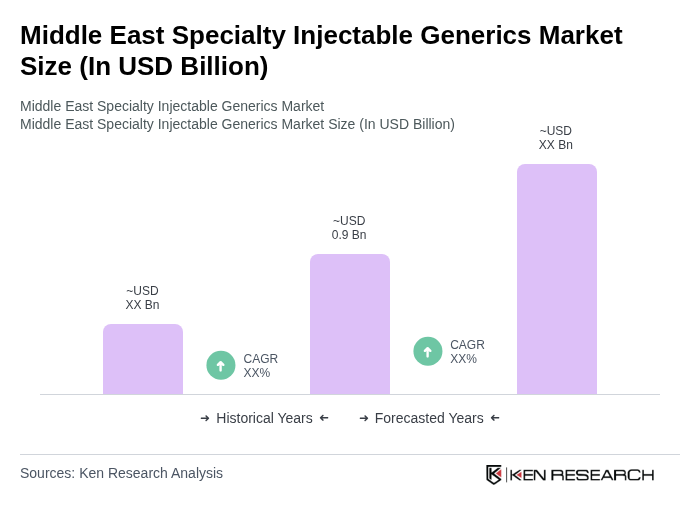
The Middle East Specialty Injectable Generics Market is valued at approximately USD 0.9 billion, reflecting a significant contribution from the broader Middle East and Africa generic injectables segment, driven by rising healthcare expenditures and the prevalence of chronic diseases.