About the Report
Base Year 2024Indonesia Generic Pharmaceuticals Market Overview
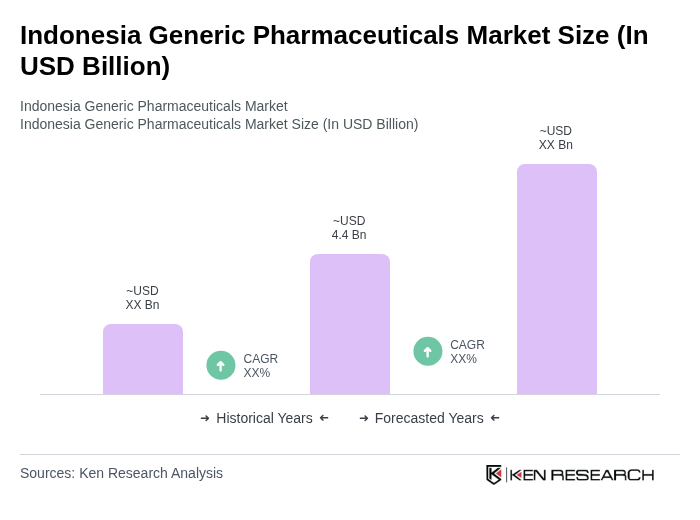
- The Indonesia Generic Pharmaceuticals Market is valued at USD 4.4 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases, rising healthcare expenditure, and the government's push for affordable medication through schemes such as the national health insurance program Jaminan Kesehatan Nasional (JKN), which has expanded access to cost-effective generic medicines. The demand for generic drugs has surged as they provide cost-effective alternatives to branded medications, making healthcare more accessible to the population, and generics account for a substantial share of total pharmaceutical volume in Indonesia.
- Key cities such as Jakarta, Surabaya, and Bandung dominate the market due to their large populations and well-established healthcare infrastructure. Jakarta, being the capital, serves as a hub for pharmaceutical companies, distribution networks, and tertiary healthcare services, while major urban centers on Java, including Surabaya and Bandung, host significant hospitals, clinics, and pharmacies that cater to the growing demand for generic pharmaceuticals. These cities also benefit from better access to specialists and higher diagnosis rates for chronic diseases, which further supports the uptake of generic medicines in both public and private sectors.
- The Indonesian government has strengthened policies that require public healthcare facilities to prioritize the use and procurement of generic medicines covered under the national formulary, particularly within the JKN program, in order to maximize budget efficiency and improve access to essential medicines. A key legal basis is the National Social Security System (Sistem Jaminan Sosial Nasional) and its implementing regulations under the Jaminan Kesehatan Nasional framework, complemented by Ministry of Health Decree No. HK.02.02/MENKES/523/2015 on the National Formulary (e?Fornas), which mandates government-funded health facilities to prioritize listed generic medicines in procurement and prescribing for JKN patients. These instruments drive higher utilization of generics by defining reimbursement rules, setting price ceilings, and requiring the use of formulary generics as first-line options in public facilities.
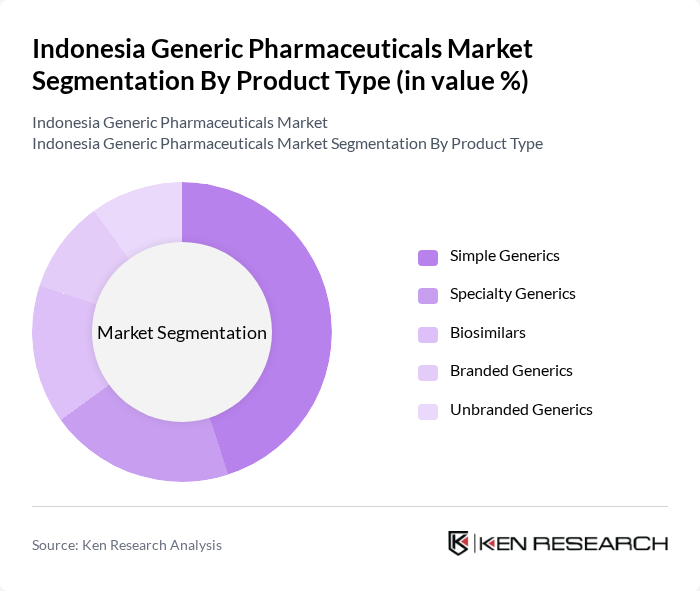
Indonesia Generic Pharmaceuticals Market Segmentation
By Product Type:The product type segmentation includes various categories such as Simple Generics, Specialty Generics, Biosimilars, Branded Generics, and Unbranded Generics. Among these, Simple Generics dominate the market due to their widespread acceptance and lower costs, making them the preferred choice for both healthcare providers and patients, especially within public reimbursement schemes. The increasing focus on cost-effective healthcare solutions, reinforced by JKN reimbursement policies and price controls on essential medicines, has led to a significant rise in the consumption of Simple Generics, particularly in urban areas where healthcare access is more prevalent and diagnosis rates for chronic diseases are higher.
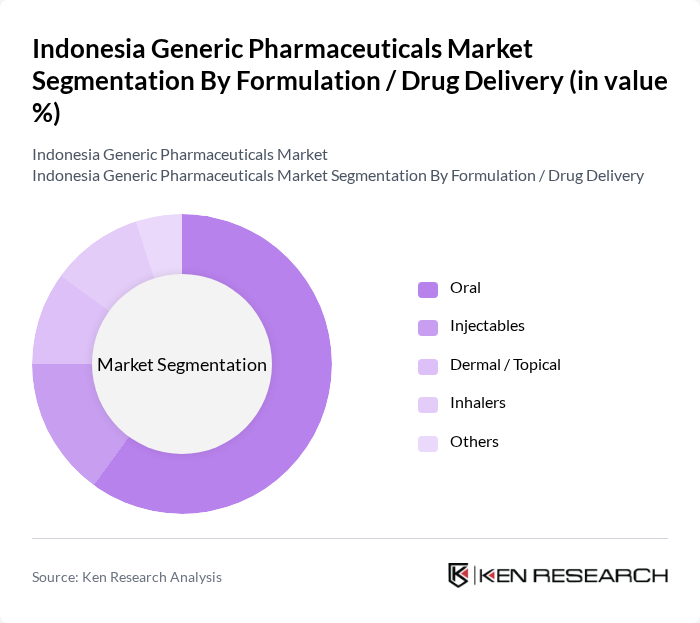
By Formulation / Drug Delivery:The formulation segmentation encompasses Oral, Injectables, Dermal/Topical, Inhalers, and Others. The Oral segment is the most dominant due to the convenience and ease of administration associated with oral medications, which aligns with prescription patterns in high-volume therapeutic areas such as diabetes, cardiovascular, and anti?infectives where tablets and capsules are the primary dosage forms. This preference is driven by consumer behavior favoring non-invasive treatment options, strong retail pharmacy distribution, and the suitability of oral generics for large-scale public procurement under JKN, leading to a higher demand for oral generic drugs in the market.
Indonesia Generic Pharmaceuticals Market Competitive Landscape
The Indonesia Generic Pharmaceuticals Market is characterized by a dynamic mix of regional and international players. Leading participants such as PT Kalbe Farma Tbk, PT Kimia Farma Tbk, PT Indofarma Tbk, PT Sanbe Farma, PT Dexa Medica, PT Soho Global Health Tbk, PT Darya?Varia Laboratoria Tbk, PT Sido Muncul Tbk, PT Bintang Toedjoe, PT Phapros Tbk, PT Enseval Putera Megatrading Tbk, PT Otto Pharmaceutical Industries, PT Harsen Laboratories, Pfizer Indonesia, and Novartis Indonesia contribute to innovation, geographic expansion, and service delivery in this space, with domestic manufacturers playing a central role in supplying generics for the JKN program and private markets.
Indonesia Generic Pharmaceuticals Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The rise in chronic diseases such as diabetes and hypertension is a significant growth driver for the generic pharmaceuticals market in Indonesia. In future, approximately 20 million Indonesians are projected to be living with diabetes, reflecting a substantial increase from previous estimates. This growing patient population necessitates affordable medication options, thereby boosting the demand for generic drugs, which are often priced around 30% lower than their branded counterparts, making them more accessible to the public.
- Government Initiatives to Promote Generic Drugs:The Indonesian government has implemented various initiatives to encourage the use of generic medications. In future, the government is expected to continue allocating several trillion Indonesian rupiah to support the production and distribution of generics. This funding aims to enhance public health by ensuring that essential medicines are available at lower costs, thereby increasing the market share of generics, which has been estimated to account for roughly half or more of total pharmaceutical volume dispensed under the national health insurance scheme.
- Rising Healthcare Expenditure:Indonesia's healthcare expenditure is projected to continue rising in future, supported by higher government spending and private sector investments, with total health expenditure currently estimated at around 3 percent of gross domestic product. This rise in expenditure is expected to enhance healthcare infrastructure and access to medications, including generics. As more healthcare facilities are established, the demand for affordable generic drugs will likely increase, further propelling market growth and improving health outcomes for the population.
Market Challenges
- Price Competition from Branded Pharmaceuticals:The Indonesian generic pharmaceuticals market faces intense price competition from established branded drugs. In future, branded pharmaceuticals are expected to account for a substantial share of the market by value, often leading to price pressures that can undermine the profitability of generic manufacturers. This competition can deter investment in generic drug development, as companies struggle to maintain margins while offering lower-priced alternatives to consumers.
- Regulatory Hurdles and Compliance Issues:Navigating the regulatory landscape in Indonesia poses significant challenges for generic pharmaceutical companies. In future, the Ministry of Health and the National Agency of Drug and Food Control are expected to continue enforcing stringent compliance measures, requiring all generics to undergo rigorous quality, safety, and efficacy evaluations. This could lead to delays in product launches and increased operational costs, which may hinder the growth of the generic market and limit the availability of affordable medications for patients.
Indonesia Generic Pharmaceuticals Market Future Outlook
The future of the Indonesian generic pharmaceuticals market appears promising, driven by increasing healthcare access and government support. As the population ages and chronic diseases become more prevalent, the demand for affordable medications will continue to rise. Additionally, advancements in technology and distribution channels, such as e-pharmacies, are expected to enhance accessibility. The focus on local manufacturing and quality assurance will further strengthen the market, ensuring that generics remain a viable option for healthcare providers and patients alike.
Market Opportunities
- Growth in E-Pharmacy Platforms:The rise of e-pharmacy platforms in Indonesia presents a significant opportunity for generic drug manufacturers. With tens of millions of internet users engaging in online shopping and digital transactions, these platforms can facilitate easier access to affordable medications, particularly in urban areas. This trend is expected to enhance the visibility and sales of generic drugs, catering to a tech-savvy population seeking convenience in healthcare.
- Expansion into Rural Markets:Expanding into rural markets offers a substantial opportunity for growth in the generic pharmaceuticals sector. Approximately 55–60 percent of Indonesia's population resides in rural areas, where access to healthcare and affordable medications is relatively limited. By establishing distribution networks and partnerships with local healthcare providers, generic manufacturers can tap into this underserved market, improving health outcomes and increasing their market share significantly.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Simple Generics Specialty Generics Biosimilars Branded Generics Unbranded Generics |
| By Formulation / Drug Delivery | Oral Injectables Dermal / Topical Inhalers Others |
| By Therapy Area | Central Nervous System Cardiovascular Respiratory Dermatology Genitourinary / Hormonal Rheumatology Diabetes Oncology Infectious Diseases Others |
| By Distribution Channel | Retail Pharmacies Hospital Pharmacies Online / E?pharmacies Wholesalers / Distributors Others |
| By End User | Public Hospitals Private Hospitals Clinics & Primary Care Centers Retail Pharmacies & Drug Stores Government Procurement Agencies (e.g., BPJS, MoH) Others |
| By Region | Java Sumatra Bali and Nusa Tenggara Kalimantan Sulawesi Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., National Agency of Drug and Food Control)
Manufacturers and Producers
Distributors and Retailers
Pharmaceutical Wholesalers
Healthcare Providers and Institutions
Industry Associations (e.g., Indonesian Pharmaceutical Association)
Financial Institutions
Players Mentioned in the Report:
PT Kalbe Farma Tbk
PT Kimia Farma Tbk
PT Indofarma Tbk
PT Sanbe Farma
PT Dexa Medica
PT Soho Global Health Tbk
PT DaryaVaria Laboratoria Tbk
PT Sido Muncul Tbk
PT Bintang Toedjoe
PT Phapros Tbk
PT Enseval Putera Megatrading Tbk
PT Otto Pharmaceutical Industries
PT Harsen Laboratories
Pfizer Indonesia
Novartis Indonesia
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Indonesia Generic Pharmaceuticals Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Indonesia Generic Pharmaceuticals Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Indonesia Generic Pharmaceuticals Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of chronic diseases
3.1.2 Government initiatives to promote generic drugs
3.1.3 Rising healthcare expenditure
3.1.4 Expanding distribution networks
3.2 Market Challenges
3.2.1 Price competition from branded pharmaceuticals
3.2.2 Regulatory hurdles and compliance issues
3.2.3 Limited awareness among healthcare providers
3.2.4 Supply chain disruptions
3.3 Market Opportunities
3.3.1 Growth in e-pharmacy platforms
3.3.2 Expansion into rural markets
3.3.3 Collaborations with healthcare providers
3.3.4 Development of biosimilars
3.4 Market Trends
3.4.1 Increasing adoption of telemedicine
3.4.2 Shift towards personalized medicine
3.4.3 Focus on sustainability in production
3.4.4 Growth of online pharmacies
3.5 Government Regulation
3.5.1 Implementation of price caps on generics
3.5.2 Mandatory registration of generic drugs
3.5.3 Quality assurance regulations
3.5.4 Incentives for local manufacturing
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Indonesia Generic Pharmaceuticals Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Indonesia Generic Pharmaceuticals Market Segmentation
8.1 By Product Type
8.1.1 Simple Generics
8.1.2 Specialty Generics
8.1.3 Biosimilars
8.1.4 Branded Generics
8.1.5 Unbranded Generics
8.2 By Formulation / Drug Delivery
8.2.1 Oral
8.2.2 Injectables
8.2.3 Dermal / Topical
8.2.4 Inhalers
8.2.5 Others
8.3 By Therapy Area
8.3.1 Central Nervous System
8.3.2 Cardiovascular
8.3.3 Respiratory
8.3.4 Dermatology
8.3.5 Genitourinary / Hormonal
8.3.6 Rheumatology
8.3.7 Diabetes
8.3.8 Oncology
8.3.9 Infectious Diseases
8.3.10 Others
8.4 By Distribution Channel
8.4.1 Retail Pharmacies
8.4.2 Hospital Pharmacies
8.4.3 Online / E?pharmacies
8.4.4 Wholesalers / Distributors
8.4.5 Others
8.5 By End User
8.5.1 Public Hospitals
8.5.2 Private Hospitals
8.5.3 Clinics & Primary Care Centers
8.5.4 Retail Pharmacies & Drug Stores
8.5.5 Government Procurement Agencies (e.g., BPJS, MoH)
8.5.6 Others
8.6 By Region
8.6.1 Java
8.6.2 Sumatra
8.6.3 Bali and Nusa Tenggara
8.6.4 Kalimantan
8.6.5 Sulawesi
8.6.6 Others
9. Indonesia Generic Pharmaceuticals Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Ownership Type (Local, MNC, JV)
9.2.3 Total Revenue (Latest Financial Year, USD / IDR)
9.2.4 Generic Pharmaceuticals Revenue Share (%)
9.2.5 3?Year Revenue CAGR (%)
9.2.6 EBITDA Margin (%)
9.2.7 Market Share in Indonesia Generics (%)
9.2.8 Volume Share in Key Therapy Areas (%)
9.2.9 Average Realized Price vs Market Index (%)
9.2.10 R&D Spend as % of Sales
9.2.11 Capex as % of Sales
9.2.12 Manufacturing Capacity Utilization (%)
9.2.13 Tender Win Rate in Public Procurement (%)
9.2.14 Geographic Coverage (No. of Provinces Served)
9.2.15 Number of SKUs in Generics Portfolio
9.2.16 Time?to?Market for New Launches (Months)
9.2.17 Days Sales Outstanding (DSO)
9.2.18 Inventory Days
9.2.19 Pharmacovigilance / Quality Incidents (No. per Year)
9.2.20 Brand Awareness Score (Physician / Pharmacist Surveys)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 PT Kalbe Farma Tbk
9.5.2 PT Kimia Farma Tbk
9.5.3 PT Indofarma Tbk
9.5.4 PT Sanbe Farma
9.5.5 PT Dexa Medica
9.5.6 PT Soho Global Health Tbk
9.5.7 PT Darya?Varia Laboratoria Tbk
9.5.8 PT Sido Muncul Tbk
9.5.9 PT Bintang Toedjoe
9.5.10 PT Phapros Tbk
9.5.11 PT Enseval Putera Megatrading Tbk
9.5.12 PT Otto Pharmaceutical Industries
9.5.13 PT Harsen Laboratories
9.5.14 Pfizer Indonesia
9.5.15 Novartis Indonesia
10. Indonesia Generic Pharmaceuticals Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Industry
10.1.4 Ministry of Trade
10.1.5 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Pharmaceutical Supply Chain Investments
10.2.3 Technology Upgrades
10.2.4 Training and Development
10.2.5 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Clinics
10.3.3 Pharmacies
10.3.4 Homecare Providers
10.3.5 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs
10.4.3 Infrastructure Readiness
10.4.4 Financial Readiness
10.4.5 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 ROI Measurement Techniques
10.5.2 Use Case Success Stories
10.5.3 Expansion Opportunities
10.5.4 Feedback Mechanisms
10.5.5 Others
11. Indonesia Generic Pharmaceuticals Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from the Indonesian Ministry of Health and pharmaceutical associations
- Review of market trends and forecasts from reputable healthcare market research publications
- Examination of regulatory frameworks and guidelines from BPOM (National Agency of Drug and Food Control)
Primary Research
- Interviews with key opinion leaders in the pharmaceutical sector, including pharmacists and healthcare professionals
- Surveys conducted with pharmaceutical manufacturers and distributors to gather insights on market dynamics
- Focus group discussions with patients and healthcare providers to understand preferences and perceptions of generic drugs
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including sales data and market share reports
- Triangulation of qualitative insights from interviews with quantitative data from surveys
- Sanity checks through expert panel reviews comprising industry veterans and academic researchers
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total pharmaceutical market size in Indonesia, with a focus on the generic segment
- Analysis of government healthcare expenditure and its impact on generic drug adoption
- Segmentation of the market by therapeutic categories and distribution channels
Bottom-up Modeling
- Collection of sales data from leading generic pharmaceutical companies operating in Indonesia
- Estimation of market share based on production volumes and sales figures
- Calculation of average pricing for generic drugs across various therapeutic classes
Forecasting & Scenario Analysis
- Development of market forecasts using historical growth rates and emerging trends in healthcare
- Scenario analysis based on potential regulatory changes and shifts in consumer behavior
- Creation of baseline, optimistic, and pessimistic projections for the next five years
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Manufacturers | 60 | Production Managers, Regulatory Affairs Specialists |
| Healthcare Providers | 80 | Pharmacists, General Practitioners |
| Patients Using Generic Drugs | 120 | Chronic Disease Patients, General Consumers |
| Distributors and Wholesalers | 60 | Supply Chain Managers, Sales Directors |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
Frequently Asked Questions
What is the current value of the Indonesia Generic Pharmaceuticals Market?
The Indonesia Generic Pharmaceuticals Market is valued at approximately USD 4.4 billion, driven by the increasing prevalence of chronic diseases, rising healthcare expenditure, and government initiatives promoting affordable medication through programs like Jaminan Kesehatan Nasional (JKN).