About the Report
Base Year 2024Saudi Arabia Recombinant Protein Manufacturing Services Market Overview
- The Saudi Arabia Recombinant Protein Manufacturing Services Market is valued at USD 20 million, based on a five-year historical analysis and alignment with the reported size and trajectory of the recombinant proteins manufacturing services market in Saudi Arabia within the broader Middle East and global context. This growth is primarily driven by the increasing demand for biopharmaceuticals, rising adoption of biologics within the Saudi Arabia biologics market, advancements in biotechnology platforms, and a growing focus on personalized and targeted therapies. The rise in chronic diseases such as cancer, diabetes, and cardiovascular disorders and the need for innovative protein-based therapies and advanced biologic products have further propelled the market, making it an increasingly important component of the healthcare and life sciences sector.
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their robust healthcare infrastructure, concentration of leading pharmaceutical and healthcare companies, and strong government support for biotechnology and biomanufacturing initiatives under national strategies such as Vision 2030 and the National Biotechnology Strategy. Riyadh, being the capital, serves as a hub for research and development, regulatory bodies, and large tertiary hospitals, while Jeddah and Dammam benefit from their strategic locations as major commercial and logistics gateways with access to international markets and industrial zones that facilitate pharmaceutical manufacturing and distribution.
- In 2023, the Saudi Food and Drug Authority (SFDA) continued to strengthen its regulatory framework for biologics and recombinant protein–based products through binding instruments such as the SFDA Guidance on Stability Studies of Biologics and Biosimilar Products issued by the Saudi Food and Drug Authority and updates to the SFDA Good Manufacturing Practice (GMP) for Medicinal Products guidelines framed under the Pharmaceutical Products Law. These regulations require all manufacturers of biologics and recombinant protein therapeutics to comply with stringent GMP standards, implement validated quality systems, and conduct comprehensive quality, safety, and stability assessments (including comparability and characterization data for biological products) before product approval, ensuring that only safe, effective, and consistently manufactured therapies reach the market.
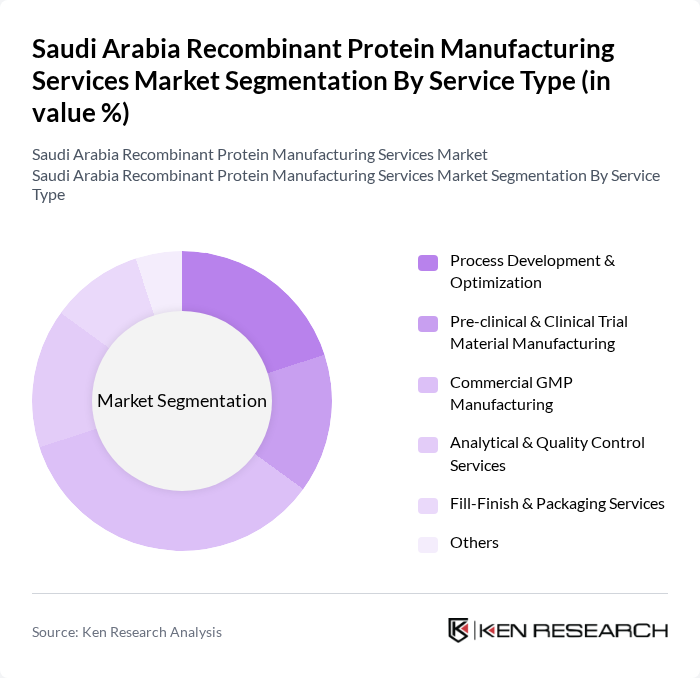
Saudi Arabia Recombinant Protein Manufacturing Services Market Segmentation
By Service Type:The service type segmentation includes various offerings that cater to the needs of the recombinant protein manufacturing process and to biopharmaceutical developers operating in Saudi Arabia and internationally. Commercial GMP Manufacturing remains the dominant sub-segment, as it is crucial for ensuring that clinical and commercial products meet SFDA and international regulatory standards and are suitable for market release. This segment is driven by the increasing demand for high-quality biopharmaceuticals, including monoclonal antibodies, hormones, enzymes, and vaccines, and the need for compliance with stringent GMP and quality guidelines adopted by the SFDA in line with ICH and WHO standards. Other significant segments include Process Development & Optimization and Analytical & Quality Control Services, which support upstream and downstream process design, scale-up, characterization, and release testing, and are increasingly important as local players and CDMOs focus on higher titers, process robustness, and cost efficiency.
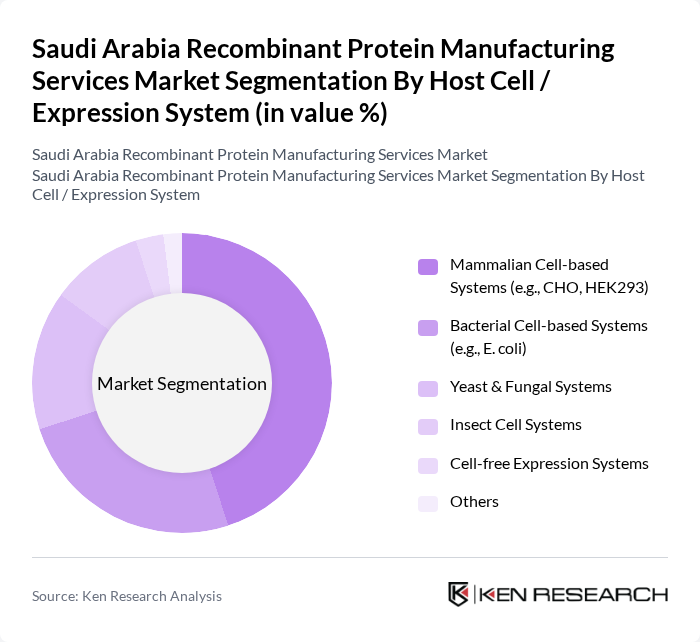
By Host Cell / Expression System:This segmentation focuses on the various host cell systems used in recombinant protein production, which broadly mirrors global technological preferences adapted to local requirements. The leading sub-segment is Mammalian Cell-based Systems, particularly CHO and HEK293 cells, which are preferred for their ability to produce complex therapeutic proteins and antibodies with human-like post-translational modifications and acceptable safety and immunogenicity profiles. Bacterial Cell-based Systems, such as E. coli, also hold a significant share due to their cost-effectiveness, ease of genetic manipulation, and rapid production capabilities, especially for non-glycosylated proteins, enzymes, and research-grade materials. Yeast & Fungal Systems and Insect Cell Systems are gaining traction as alternatives for specific applications that require scalable production with certain post-translational modifications or where cost and yield considerations are critical, while Cell-free Expression Systems are emerging for rapid prototyping, high-throughput screening, and specialized research needs.
Saudi Arabia Recombinant Protein Manufacturing Services Market Competitive Landscape
The Saudi Arabia Recombinant Protein Manufacturing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Saudi Arabian Japanese Pharmaceutical Company Ltd. (SAJAPHCO), Saudi Pharmaceutical Industries & Medical Appliances Corporation (SPIMACO), Jamjoom Pharma, Tabuk Pharmaceuticals Manufacturing Company, Riyadh Pharma (Riyadh Pharmaceuticals), Banaja Holding Company, Tamer Group, Saudi Chemical Company Holding – Pharma Sector (including AJA Pharma), Modern Pharmaceutical Company (MPC), Hikma Pharmaceuticals – Saudi Operations, Jamjoom Medical Industries, Sudair Pharma Company, SPIMACO Addwaeh Biotech Subsidiaries, Local CDMOs / CMOs Focused on Biologics & Recombinant Proteins, Selected International CDMOs with Active Presence / Partnerships in Saudi Arabia contribute to innovation, geographic expansion, localization of biologics manufacturing, and service delivery in this space as the country moves to expand domestic biopharmaceutical capabilities.
Saudi Arabia Recombinant Protein Manufacturing Services Market Industry Analysis
Growth Drivers
- Increasing Demand for Biopharmaceuticals:The biopharmaceutical market in Saudi Arabia is projected to reach approximately SAR 25 billion by 2025, driven by a growing population and rising healthcare needs. The demand for innovative therapies, particularly in oncology and autoimmune diseases, is propelling the recombinant protein manufacturing sector. With over 70% of the population suffering from chronic diseases, the need for effective biopharmaceuticals is critical, fostering growth in this market segment.
- Advancements in Biotechnology:Saudi Arabia's investment in biotechnology has surged, with the government allocating SAR 2 billion for research and development in future. This funding supports the development of advanced recombinant protein technologies, enhancing production efficiency and product quality. The establishment of biotechnology parks and incubators is fostering innovation, enabling local companies to compete globally and meet the increasing demand for biopharmaceuticals.
- Government Support and Funding:The Saudi government is actively promoting the biotechnology sector through initiatives like Vision 2030, which aims to diversify the economy. In future, the government plans to increase funding for biotech projects by 30%, facilitating the growth of recombinant protein manufacturing services. This support includes tax incentives and grants, encouraging private sector investment and fostering a conducive environment for industry expansion.
Market Challenges
- High Production Costs:The cost of producing recombinant proteins in Saudi Arabia is significantly high, with estimates reaching SAR 600 million for large-scale production facilities. This is primarily due to the expensive raw materials and advanced technologies required. As a result, local manufacturers face challenges in pricing their products competitively against international players, which can hinder market growth and limit access to affordable biopharmaceuticals.
- Regulatory Hurdles:The regulatory landscape for biopharmaceuticals in Saudi Arabia is complex, with stringent requirements for product approval. The average time for regulatory approval can exceed 24 months, causing delays in bringing new products to market. This lengthy process can deter investment in recombinant protein manufacturing, as companies may seek more favorable regulatory environments in other regions, impacting local industry growth.
Saudi Arabia Recombinant Protein Manufacturing Services Market Future Outlook
The future of the recombinant protein manufacturing services market in Saudi Arabia appears promising, driven by increasing investments in healthcare infrastructure and biotechnology. The government's commitment to enhancing R&D capabilities and fostering public-private partnerships is expected to stimulate innovation. Additionally, the rising focus on personalized medicine will likely create new avenues for growth, enabling companies to develop tailored therapies that meet specific patient needs, thereby expanding market potential.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Saudi government is investing SAR 70 billion in healthcare infrastructure in future, which will enhance access to biopharmaceuticals. This expansion will create opportunities for recombinant protein manufacturers to establish partnerships with healthcare providers, ensuring a steady demand for innovative therapies and improving patient outcomes.
- Collaborations with Research Institutions:Collaborations between biotech firms and research institutions are on the rise, with over 40 active partnerships reported in future. These collaborations facilitate knowledge transfer and innovation, enabling companies to leverage cutting-edge research in recombinant protein development, thus enhancing their competitive edge in the market.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Service Type | Process Development & Optimization Pre-clinical & Clinical Trial Material Manufacturing Commercial GMP Manufacturing Analytical & Quality Control Services Fill-Finish & Packaging Services Others |
| By Host Cell / Expression System | Mammalian Cell-based Systems (e.g., CHO, HEK293) Bacterial Cell-based Systems (e.g., E. coli) Yeast & Fungal Systems Insect Cell Systems Cell-free Expression Systems Others |
| By Application | Therapeutic Protein Production Vaccine & Antigen Production Diagnostics & Assay Development Research Reagents & Tool Proteins Others |
| By Client Type | Pharmaceutical Companies Biotechnology Firms & Start-ups Academic & Research Institutions Contract Research Organizations (CROs) Government & Public Health Agencies Others |
| By Production Scale | R&D / Pre-clinical Scale Clinical Scale Commercial / Large Scale Others |
| By Mode of Service Delivery | Full-scale Contract Manufacturing (CDMO) Project-based / Modular Services Long-term Strategic Partnerships Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Biopharmaceutical Companies
Contract Manufacturing Organizations (CMOs)
Healthcare Providers and Hospitals
Pharmaceutical Distributors
Biotechnology Research Institutions
Supply Chain and Logistics Companies
Players Mentioned in the Report:
Saudi Arabian Japanese Pharmaceutical Company Ltd. (SAJAPHCO)
Saudi Pharmaceutical Industries & Medical Appliances Corporation (SPIMACO)
Jamjoom Pharma
Tabuk Pharmaceuticals Manufacturing Company
Riyadh Pharma (Riyadh Pharmaceuticals)
Banaja Holding Company
Tamer Group
Saudi Chemical Company Holding Pharma Sector (including AJA Pharma)
Modern Pharmaceutical Company (MPC)
Hikma Pharmaceuticals Saudi Operations
Jamjoom Medical Industries
Sudair Pharma Company
SPIMACO Addwaeh Biotech Subsidiaries
Local CDMOs / CMOs Focused on Biologics & Recombinant Proteins
Selected International CDMOs with Active Presence / Partnerships in Saudi Arabia
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Recombinant Protein Manufacturing Services Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Recombinant Protein Manufacturing Services Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Recombinant Protein Manufacturing Services Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for biopharmaceuticals
3.1.2 Advancements in biotechnology
3.1.3 Government support and funding
3.1.4 Rising prevalence of chronic diseases
3.2 Market Challenges
3.2.1 High production costs
3.2.2 Regulatory hurdles
3.2.3 Limited skilled workforce
3.2.4 Competition from established markets
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Collaborations with research institutions
3.3.3 Growth in personalized medicine
3.3.4 Increasing investments in R&D
3.4 Market Trends
3.4.1 Shift towards sustainable manufacturing
3.4.2 Integration of AI in protein production
3.4.3 Focus on biosimilars
3.4.4 Rise of contract manufacturing organizations (CMOs)
3.5 Government Regulation
3.5.1 Regulatory frameworks for biopharmaceuticals
3.5.2 Quality assurance standards
3.5.3 Intellectual property protections
3.5.4 Import/export regulations for biotech products
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Recombinant Protein Manufacturing Services Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Recombinant Protein Manufacturing Services Market Segmentation
8.1 By Service Type
8.1.1 Process Development & Optimization
8.1.2 Pre-clinical & Clinical Trial Material Manufacturing
8.1.3 Commercial GMP Manufacturing
8.1.4 Analytical & Quality Control Services
8.1.5 Fill-Finish & Packaging Services
8.1.6 Others
8.2 By Host Cell / Expression System
8.2.1 Mammalian Cell-based Systems (e.g., CHO, HEK293)
8.2.2 Bacterial Cell-based Systems (e.g., E. coli)
8.2.3 Yeast & Fungal Systems
8.2.4 Insect Cell Systems
8.2.5 Cell-free Expression Systems
8.2.6 Others
8.3 By Application
8.3.1 Therapeutic Protein Production
8.3.2 Vaccine & Antigen Production
8.3.3 Diagnostics & Assay Development
8.3.4 Research Reagents & Tool Proteins
8.3.5 Others
8.4 By Client Type
8.4.1 Pharmaceutical Companies
8.4.2 Biotechnology Firms & Start-ups
8.4.3 Academic & Research Institutions
8.4.4 Contract Research Organizations (CROs)
8.4.5 Government & Public Health Agencies
8.4.6 Others
8.5 By Production Scale
8.5.1 R&D / Pre-clinical Scale
8.5.2 Clinical Scale
8.5.3 Commercial / Large Scale
8.5.4 Others
8.6 By Mode of Service Delivery
8.6.1 Full-scale Contract Manufacturing (CDMO)
8.6.2 Project-based / Modular Services
8.6.3 Long-term Strategic Partnerships
8.6.4 Others
8.7 By Region
8.7.1 Central Region
8.7.2 Eastern Region
8.7.3 Western Region
8.7.4 Southern Region
9. Saudi Arabia Recombinant Protein Manufacturing Services Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Saudi Arabia Recombinant Protein Manufacturing Services Revenue (US$ Mn)
9.2.4 3-year Revenue CAGR in Saudi Arabia (%)
9.2.5 EBITDA Margin (%)
9.2.6 Capacity Utilization Rate (%)
9.2.7 Average Batch Success Rate (%)
9.2.8 On-time Delivery Rate (%)
9.2.9 R&D Spend as % of Total Revenue
9.2.10 Number of Active CDMO / CMO Contracts
9.2.11 GMP-certified Manufacturing Sites (Count)
9.2.12 Customer Retention / Renewal Rate (%)
9.2.13 Average Contract Tenure (Years)
9.2.14 Compliance / Audit Findings per Site (Annual Avg.)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Saudi Arabian Japanese Pharmaceutical Company Ltd. (SAJAPHCO)
9.5.2 Saudi Pharmaceutical Industries & Medical Appliances Corporation (SPIMACO)
9.5.3 Jamjoom Pharma
9.5.4 Tabuk Pharmaceuticals Manufacturing Company
9.5.5 Riyadh Pharma (Riyadh Pharmaceuticals)
9.5.6 Banaja Holding Company
9.5.7 Tamer Group
9.5.8 Saudi Chemical Company Holding – Pharma Sector (including AJA Pharma)
9.5.9 Modern Pharmaceutical Company (MPC)
9.5.10 Hikma Pharmaceuticals – Saudi Operations
9.5.11 Jamjoom Medical Industries
9.5.12 Sudair Pharma Company
9.5.13 SPIMACO Addwaeh Biotech Subsidiaries
9.5.14 Local CDMOs / CMOs Focused on Biologics & Recombinant Proteins
9.5.15 Selected International CDMOs with Active Presence / Partnerships in Saudi Arabia
10. Saudi Arabia Recombinant Protein Manufacturing Services Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Industry and Mineral Resources
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Pharmaceutical Sector Investments
10.2.2 Biotechnology Sector Investments
10.2.3 Research and Development Investments
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Pharmaceutical Companies
10.3.2 Biotechnology Firms
10.3.3 Research Institutions
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training and Support Needs
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 ROI Measurement Techniques
10.5.2 Use Case Development
10.5.3 Long-term Sustainability Considerations
10.5.4 Others
11. Saudi Arabia Recombinant Protein Manufacturing Services Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from biotechnology associations and government publications
- Review of scientific literature and patents related to recombinant protein technologies
- Examination of market trends and forecasts from reputable market research firms
Primary Research
- Interviews with key opinion leaders in the biotechnology and pharmaceutical sectors
- Surveys targeting R&D managers and production heads in recombinant protein manufacturing
- Focus groups with industry experts to gather insights on market dynamics and challenges
Validation & Triangulation
- Cross-validation of findings through multiple data sources including trade publications and expert interviews
- Triangulation of quantitative data with qualitative insights from industry stakeholders
- Sanity checks through peer reviews and expert panel discussions to ensure data reliability
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total market size based on national healthcare expenditure and biotechnology investments
- Segmentation of the market by application areas such as therapeutics, diagnostics, and research
- Incorporation of government initiatives and funding in biotechnology to assess growth potential
Bottom-up Modeling
- Collection of production data from leading recombinant protein manufacturers in Saudi Arabia
- Cost analysis based on operational expenses and pricing strategies of key players
- Volume estimates derived from production capacity and market demand forecasts
Forecasting & Scenario Analysis
- Utilization of time-series analysis to project market growth based on historical data
- Scenario modeling considering factors such as regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Applications | 120 | R&D Directors, Regulatory Affairs Managers |
| Biotechnology Research Institutions | 90 | Research Scientists, Lab Managers |
| Clinical Trials and Testing | 80 | Clinical Research Coordinators, Quality Assurance Officers |
| Industrial Manufacturing | 70 | Production Managers, Supply Chain Analysts |
| Regulatory Compliance and Policy | 60 | Compliance Officers, Policy Makers |
Frequently Asked Questions
What is the current value of the Saudi Arabia Recombinant Protein Manufacturing Services Market?
The Saudi Arabia Recombinant Protein Manufacturing Services Market is valued at approximately USD 20 million, reflecting a historical analysis and alignment with the broader Middle East and global recombinant proteins manufacturing services market trends.