Region:Middle East
Author(s):Dev
Product Code:KRAA1016
Pages:88
Published On:January 2026
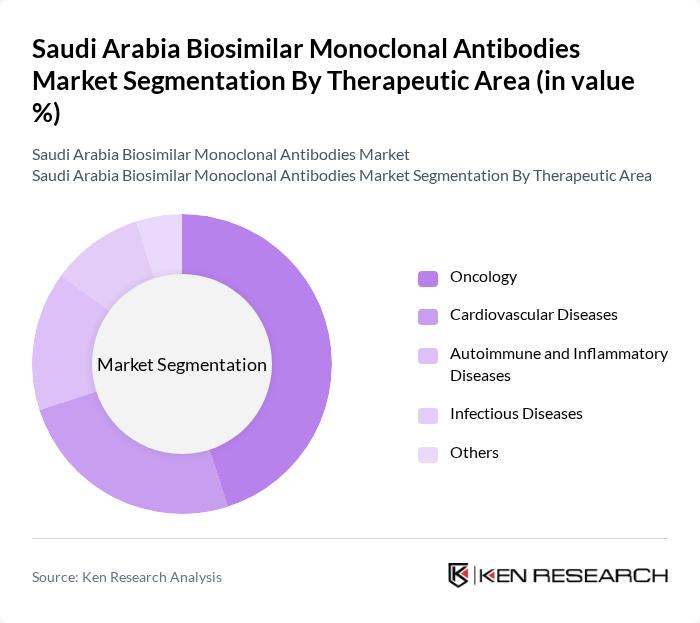
By Therapeutic Area:The therapeutic area segmentation of the market includes various categories such as Oncology, Cardiovascular Diseases, Autoimmune and Inflammatory Diseases, Infectious Diseases, and Others. Oncology is the leading segment due to the high incidence of cancer in the region, driving demand for effective treatment options. The increasing focus on personalized medicine and targeted therapies further enhances the growth of this segment.
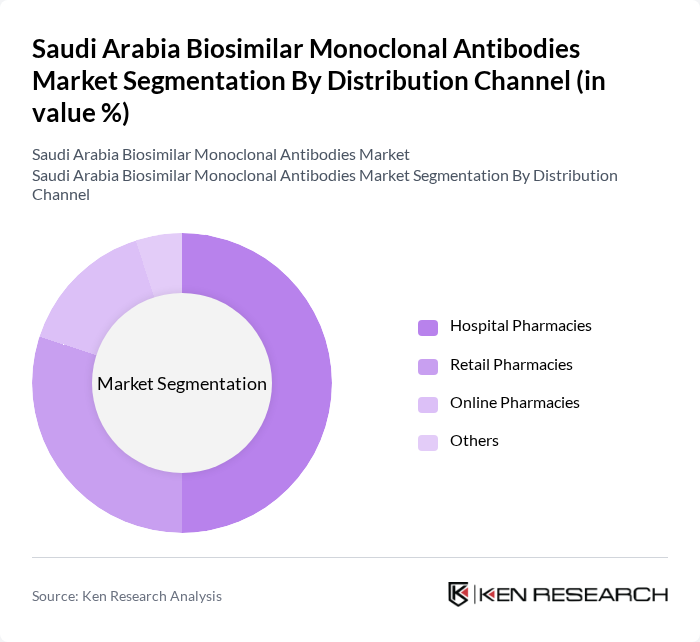
By Distribution Channel:The distribution channel segmentation includes Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, and Others. Hospital Pharmacies dominate this segment as they are the primary point of access for patients requiring specialized treatments, including biosimilars. The increasing trend of digital health and telemedicine is also contributing to the growth of online pharmacies.
The Saudi Arabia Biosimilar Monoclonal Antibodies Market is characterized by a dynamic mix of regional and international players. Leading participants such as Amgen Inc., Pfizer Inc., Roche Holding AG, Merck & Co., Inc., Novartis AG, Sanofi S.A., AbbVie Inc., Teva Pharmaceutical Industries Ltd., Mylan N.V., Celltrion Healthcare Co., Ltd., Sandoz International GmbH, Biocon Ltd., Hikma Pharmaceuticals PLC, LG Chem Ltd., Samsung Bioepis Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the biosimilar monoclonal antibodies market in Saudi Arabia appears promising, driven by increasing healthcare investments and a growing focus on innovative treatment solutions. As the government continues to enhance healthcare infrastructure and regulatory frameworks, the adoption of biosimilars is expected to rise. Additionally, the integration of digital health technologies and personalized medicine will likely create new avenues for biosimilar development, fostering a more competitive and accessible market landscape in future.
| Segment | Sub-Segments |
|---|---|
| By Therapeutic Area | Oncology Cardiovascular Diseases Autoimmune and Inflammatory Diseases Infectious Diseases Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Others |
| By Formulation | Injectable Infusion Others |
| By End-User | Hospitals Clinics Homecare Settings Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Policy Support | Subsidies Tax Exemptions Regulatory Support Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Treatment Centers | 100 | Oncologists, Pharmacists |
| Rheumatology Clinics | 80 | Rheumatologists, Nurse Practitioners |
| Hospital Pharmacy Departments | 90 | Pharmacy Directors, Clinical Pharmacists |
| Patient Advocacy Groups | 70 | Patient Representatives, Healthcare Advocates |
| Health Insurance Providers | 60 | Policy Analysts, Medical Directors |
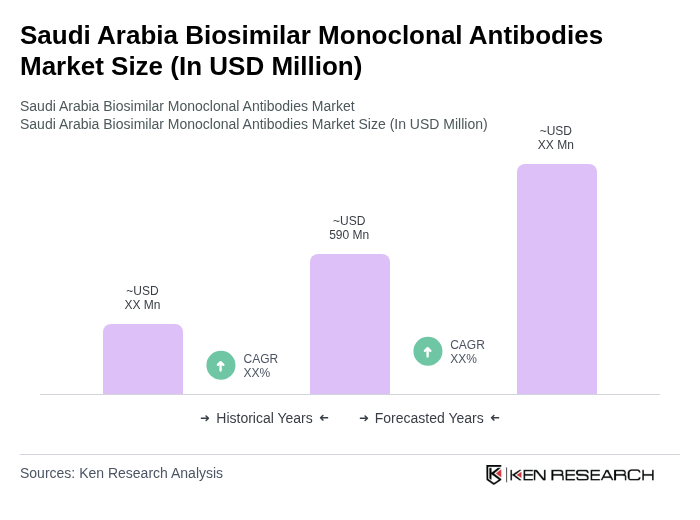
The Saudi Arabia Biosimilar Monoclonal Antibodies Market is valued at approximately USD 590 million, reflecting significant growth driven by the rising prevalence of chronic diseases and increasing healthcare expenditure.