Vietnam Generic Pharmaceuticals Market Overview
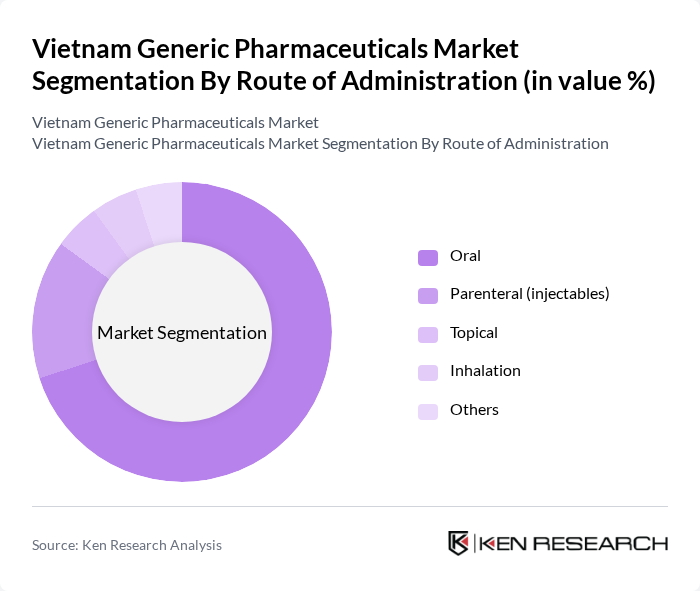
- The Vietnam Generic Pharmaceuticals Market is valued at USD 3.2 billion, based on a five-year historical analysis and recent industry estimates for the national generic drugs segment. This growth is primarily driven by the increasing demand for affordable medications, the rising prevalence of chronic and lifestyle-related diseases such as cardiovascular diseases and diabetes, and government initiatives to promote the use of generics through cost-containment and expanded health insurance coverage. The market has seen a significant shift towards generics as healthcare costs continue to rise and income and insurance coverage expand, making generic medicines a preferred choice for both healthcare providers and patients in Vietnam’s price-sensitive environment.
- Key demand and distribution hubs in this market include Ho Chi Minh City, Hanoi, and Da Nang, which dominate due to their robust healthcare infrastructure, higher population density, and greater access to healthcare services. These urban centers host a high concentration of hospitals, private clinics, and pharmacies, and are home to many domestic and multinational pharmaceutical companies, manufacturing plants, and research or distribution facilities, facilitating innovation, clinical adoption, and nationwide distribution of generic drugs and thereby enhancing their market presence.
- To enhance the quality, safety, and accessibility of pharmaceuticals, including generics, the Vietnamese government has strengthened the regulatory framework through instruments such as the Law on Pharmacy issued by the National Assembly in 2016 and Decree No. 54/2017/ND-CP issued by the Government in 2017, which detail requirements for marketing authorization, bioequivalence, good manufacturing practice (GMP), and quality control for generic medicines. Under these rules, generic drugs must comply with stringent quality assessments, GMP standards aligned with international practices, and dossier and clinical or bioequivalence requirements before being approved for sale, ensuring that patients receive safe and effective medications while supporting wider access through public procurement and reimbursement policies.
Vietnam Generic Pharmaceuticals Market Segmentation
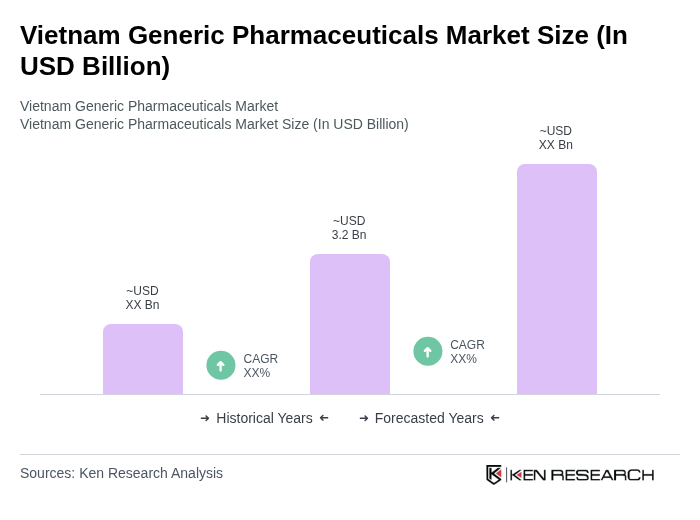
By Drug Type:The market is segmented into two primary categories: pure generic drugs and branded generics. Pure generic drugs are typically less expensive and are often preferred by cost-sensitive consumers and public hospitals seeking to optimize treatment costs in tenders and reimbursement schemes. In contrast, branded generics, which are marketed under a brand name but are chemically identical in active ingredients to originator products, appeal to consumers and physicians seeking perceived quality, consistent supply, and strong manufacturer reputation. The demand for branded generics has been increasing in urban areas due to rising consumer awareness, marketing efforts by domestic manufacturers, and trust in established brands that comply with international GMP and quality standards.
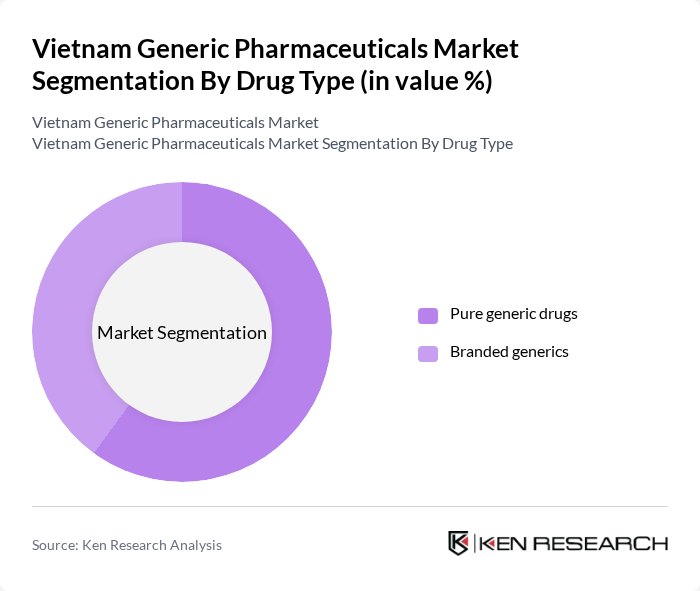
By Route of Administration:The market is further segmented by route of administration, including oral, parenteral (injectables), topical, inhalation, and others. Oral administration dominates the market due to its convenience, patient adherence, and suitability for a wide range of chronic therapies such as antihypertensives, antidiabetics, and cardiovascular drugs, which are increasingly prescribed in generic form. Parenteral administration is also significant, particularly for antibiotics, oncology medicines, and hospital-based therapies that require rapid absorption or are not suitable for oral intake, with demand supported by hospital procurement of cost-effective generic injectables. The increasing prevalence of chronic and serious conditions treated in hospital settings has led to a growing demand for injectables, while topical, inhalation, and other routes serve important but more specialized niches in dermatology, respiratory care, and emergency medicine.
Vietnam Generic Pharmaceuticals Market Competitive Landscape
The Vietnam Generic Pharmaceuticals Market is characterized by a dynamic mix of regional and international players. Leading participants such as DHG Pharmaceutical JSC, Traphaco JSC, Imexpharm Corporation, Domesco Medical Import-Export JSC, OPC Pharmaceutical JSC, Pymepharco JSC, Mekophar Chemical Pharmaceutical JSC, SPM JSC, Vimedimex Medi-Pharma JSC, Sanofi Vietnam, Bayer Vietnam, Novartis Vietnam, Pfizer Vietnam, GSK Vietnam, Abbott Vietnam contribute to innovation, geographic expansion, and service delivery in this space, with domestic firms particularly strong in cost-effective generics and over-the-counter products and multinationals focusing on higher-value and specialty portfolios.
Vietnam Generic Pharmaceuticals Market Industry Analysis
Growth Drivers
- Increasing Healthcare Expenditure:Vietnam's healthcare expenditure is projected to reach approximately $20 billion in future, reflecting a significant increase from $15 billion in past years. This growth is driven by the government's commitment to improving healthcare access and quality, which includes expanding insurance coverage. As more citizens gain access to healthcare services, the demand for affordable generic medications is expected to rise, further stimulating market growth in the generic pharmaceuticals sector.
- Rising Prevalence of Chronic Diseases:The World Health Organization reported that chronic diseases account for 77% of all deaths in Vietnam, with conditions such as diabetes and cardiovascular diseases on the rise. By future, it is estimated that over 15 million people will be living with chronic diseases, increasing the demand for effective and affordable treatment options. This trend is driving the growth of the generic pharmaceuticals market as patients seek cost-effective alternatives to branded medications.
- Government Initiatives to Promote Generics:The Vietnamese government has implemented various initiatives to promote the use of generic drugs, including the National Drug Policy, which aims to increase the availability of generics. In future, the government plans to allocate $1.5 billion to support local pharmaceutical manufacturers. These initiatives are expected to enhance the production and distribution of generics, making them more accessible to the population and driving market growth.
Market Challenges
- Intense Competition from Local and International Players:The Vietnamese generic pharmaceuticals market is characterized by intense competition, with roughly 230–250 local manufacturers and numerous international companies vying for market share. This saturation leads to price wars, which can erode profit margins. In future, the average profit margin for generic drug manufacturers is expected to decline to around 10%, making it challenging for companies to sustain profitability while maintaining quality standards.
- Regulatory Hurdles and Compliance Issues:Navigating the regulatory landscape in Vietnam can be complex, with stringent requirements for drug registration and approval. In future, it is estimated that the average time for drug approval will remain around 12-18 months, which can delay market entry for new generics. Additionally, compliance with quality control standards can be costly, posing a significant challenge for smaller manufacturers trying to compete in the market.
Vietnam Generic Pharmaceuticals Market Future Outlook
The Vietnam generic pharmaceuticals market is poised for significant growth, driven by increasing healthcare expenditure and a rising prevalence of chronic diseases. As the government continues to promote generics through supportive policies, local manufacturers are expected to enhance their production capabilities. Additionally, the adoption of digital health technologies will facilitate better access to medications. Overall, the market is likely to evolve, focusing on affordability and accessibility, while addressing regulatory challenges to ensure sustainable growth.
Market Opportunities
- Growth in E-Pharmacy and Online Sales:The e-pharmacy sector in Vietnam is projected to grow significantly, with online sales expected to reach $1 billion in future. This growth presents an opportunity for generic pharmaceutical companies to expand their reach and improve accessibility to medications, particularly in rural areas where traditional pharmacies may be limited.
- Expansion of Healthcare Infrastructure:Vietnam's healthcare infrastructure is undergoing rapid expansion, with the government investing $2 billion in new hospitals and clinics in future. This development will increase the demand for generic medications as healthcare facilities seek cost-effective solutions to treat patients, creating a favorable environment for generic pharmaceutical companies to thrive.