Region:Europe
Author(s):Shubham
Product Code:KRAB1106
Pages:93
Published On:October 2025
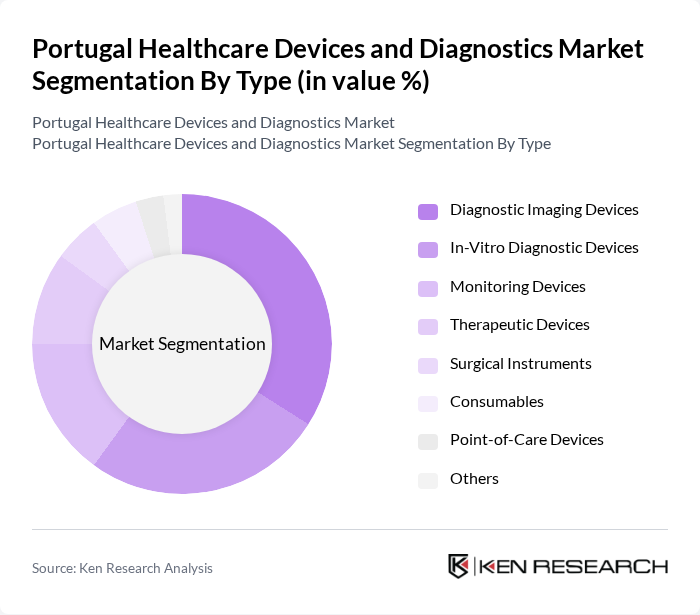
By Type:The market is segmented into various types of healthcare devices and diagnostics, including diagnostic imaging devices, in-vitro diagnostic devices, monitoring devices, therapeutic devices, surgical instruments, consumables, point-of-care devices, and others. Among these, diagnostic imaging devices are leading the market due to their critical role in disease diagnosis and management. The increasing adoption of advanced imaging technologies, such as MRI and CT scans, is driving this segment's growth. In-vitro diagnostic devices also hold a significant share, fueled by the rising demand for early disease detection and personalized medicine .
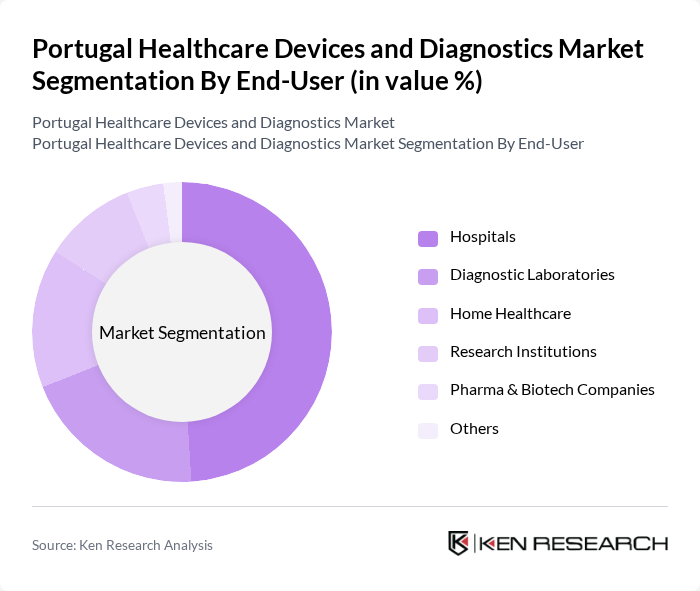
By End-User:The end-user segmentation includes hospitals, diagnostic laboratories, home healthcare, research institutions, pharma & biotech companies, and others. Hospitals are the dominant end-user segment, driven by the increasing number of healthcare facilities and the rising demand for advanced medical technologies. Diagnostic laboratories also play a crucial role, as they require sophisticated diagnostic devices to provide accurate and timely results. The home healthcare segment is gaining traction due to the growing preference for at-home monitoring and treatment options .
The Portugal Healthcare Devices and Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Siemens Healthineers, Philips Healthcare, GE Healthcare, Roche Diagnostics, Abbott Laboratories, Medtronic, Becton, Dickinson and Company, Johnson & Johnson, Stryker Corporation, Thermo Fisher Scientific, Canon Medical Systems, Olympus Corporation, Hologic, Inc., Mindray Medical International Limited, Fujifilm Holdings Corporation, Bial – Portela & Cª, S.A., LiMM Therapeutics, Werfen (Instrumentation Laboratory), Grifols S.A., Sysmex Europe GmbH contribute to innovation, geographic expansion, and service delivery in this space.
The future of the healthcare devices and diagnostics market in Portugal appears promising, driven by ongoing technological innovations and a growing emphasis on preventive healthcare. As the healthcare system increasingly adopts telemedicine and home healthcare services, the demand for advanced diagnostic tools is expected to rise. Additionally, the integration of AI and data analytics will enhance diagnostic accuracy, leading to improved patient outcomes and greater efficiency in healthcare delivery across the country.
| Segment | Sub-Segments |
|---|---|
| By Type | Diagnostic Imaging Devices In-Vitro Diagnostic Devices Monitoring Devices Therapeutic Devices Surgical Instruments Consumables Point-of-Care Devices Others |
| By End-User | Hospitals Diagnostic Laboratories Home Healthcare Research Institutions Pharma & Biotech Companies Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Retail Pharmacies Others |
| By Application | Cardiovascular Diabetes Management Oncology Infectious Disease Neurology Others |
| By Price Range | Low-End Devices Mid-Range Devices High-End Devices |
| By Technology | Digital Health Technologies Molecular Diagnostics (PCR, dPCR, etc.) Traditional Technologies Hybrid Technologies |
| By Regulatory Compliance | CE Marked Devices FDA Approved Devices ISO Certified Devices Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Diagnostic Imaging Devices | 60 | Radiologists, Imaging Technicians |
| Surgical Instruments | 50 | Surgeons, Operating Room Managers |
| Patient Monitoring Systems | 45 | Nurses, Clinical Engineers |
| Wearable Health Devices | 40 | Healthcare Innovators, Product Managers |
| Laboratory Diagnostic Equipment | 40 | Lab Technicians, Quality Control Managers |
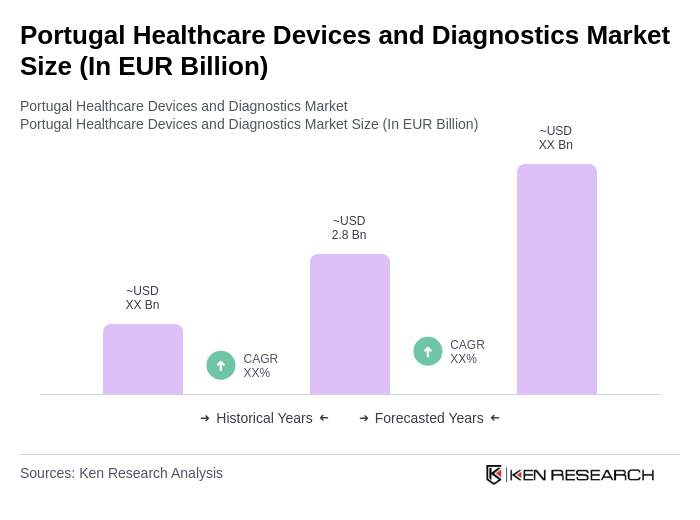
The Portugal Healthcare Devices and Diagnostics Market is valued at approximately EUR 2.8 billion, driven by factors such as an aging population, increasing chronic diseases, and advancements in medical technology.