About the Report
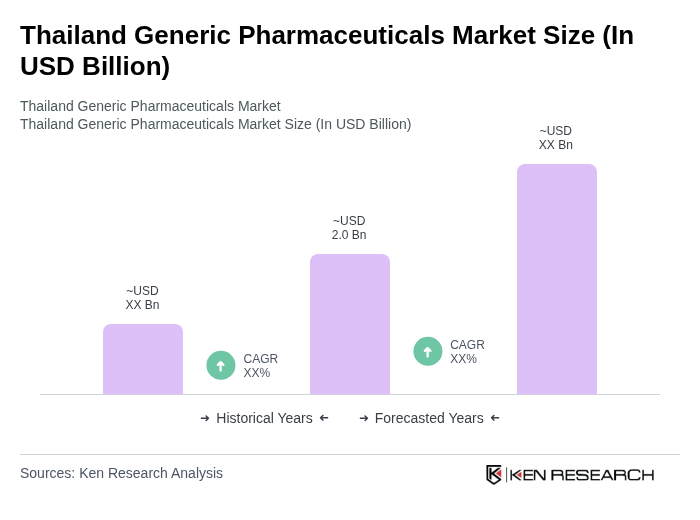
Base Year 2024Thailand Generic Pharmaceuticals Market Overview
- The Thailand Generic Pharmaceuticals Market is valued at USD 2.0 billion, based on a five-year historical analysis and the share of generics in Thailand’s overall pharmaceutical spending. Thailand’s total pharmaceutical market has recently been estimated at around USD 8.9 billion, with generics (hospital-prescribed and retail) accounting for a substantial share of domestic drug sales. This growth is primarily driven by the increasing demand for affordable medications, the rising prevalence of chronic non-communicable diseases such as diabetes, cardiovascular disease, and cancer, and government initiatives to promote generic drug usage through public insurance schemes. Rising healthcare utilization under universal coverage and cost-containment pressures in public hospitals have reinforced the shift towards generics as healthcare costs continue to rise, making them a more viable option for both consumers and healthcare providers.
- Key demand centers for generic pharmaceuticals in Thailand include Bangkok and other major regional hubs such as Chiang Mai and Nakhon Ratchasima, which host large public and private hospitals as well as dense networks of retail pharmacies. Bangkok, as the capital, serves as a hub for pharmaceutical companies, tertiary referral hospitals, and specialized medical centers, while Chiang Mai and Nakhon Ratchasima benefit from their strategic locations as regional medical hubs with growing healthcare infrastructure and medical tourism activity. The concentration of healthcare facilities, higher patient volumes, and better distribution logistics in these cities supports stronger penetration and uptake of generic medicines.
- The core policy framework driving generic utilization is the National Health Security Act B.E. 2545 (2002), issued by the National Assembly and implemented through the National Health Security Office (NHSO), which established the Universal Coverage Scheme and mandates that publicly financed health services prioritize cost-effective medicines, including generics, as listed in the National List of Essential Medicines (NLEM). Under this framework, reimbursement and hospital procurement are guided by the NLEM, resulting in a high proportion of generic products in public sector drug spending and improving access to essential medicines while containing the financial burden on the health system and patients.
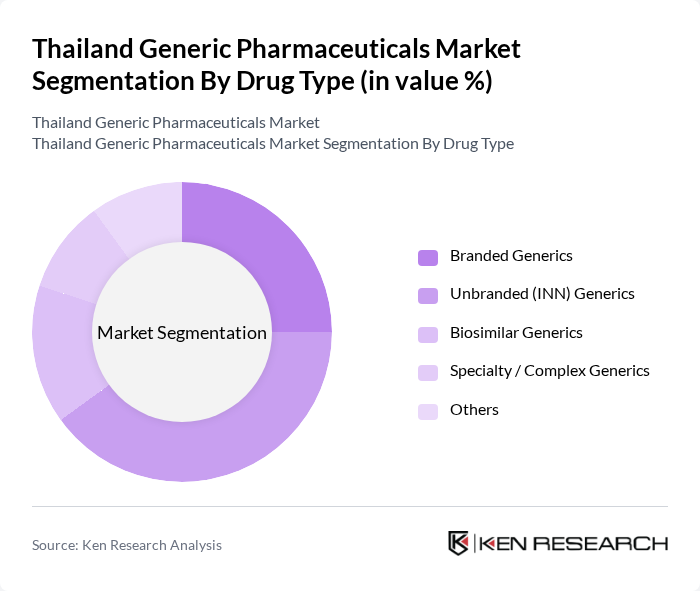
Thailand Generic Pharmaceuticals Market Segmentation
By Drug Type:The drug type segmentation includes various categories such as Branded Generics, Unbranded (INN) Generics, Biosimilar Generics, Specialty / Complex Generics, and Others. In Thailand’s hospital and public insurance–driven market, generic drugs on the National List of Essential Medicines are widely prescribed, and a large share of these are supplied as non?proprietary or institution-branded products, supporting the strong role of Unbranded (INN) Generics in overall volume. The trend towards cost-effective treatment options, tighter hospital drug budgets, and promotion of NLEM-based prescribing has led to a continued rise in the consumption of unbranded and branded generics, making them a preferred choice in the Thai healthcare system, particularly in public hospitals. Biosimilar and specialty/complex generics are still smaller but growing segments, driven by the increasing burden of oncology and autoimmune diseases and efforts to improve biologic affordability.
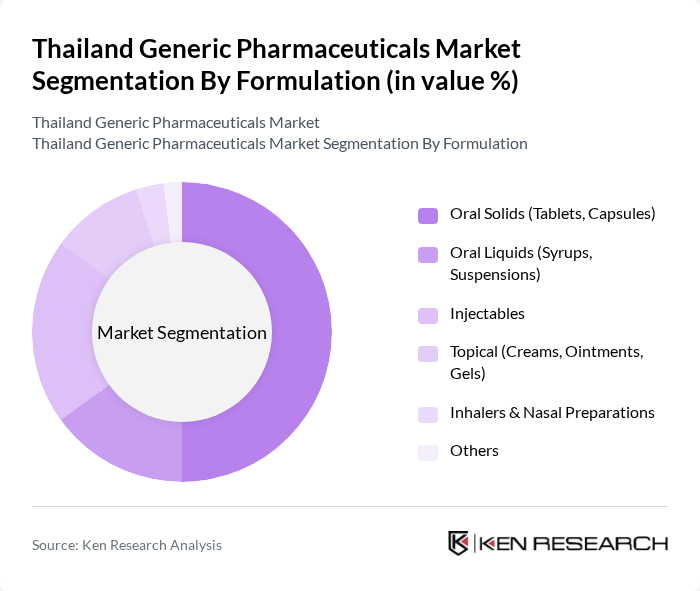
By Formulation:The formulation segmentation encompasses Oral Solids (Tablets, Capsules), Oral Liquids (Syrups, Suspensions), Injectables, Topical (Creams, Ointments, Gels), Inhalers & Nasal Preparations, and Others. Oral Solids are the leading formulation type, in line with global and regional patterns where solid dosage forms account for the majority of prescription and over-the-counter medicines. Their convenience, ease of administration, longer shelf life, and cost-effectiveness make them the preferred choice in both hospital and retail pharmacy channels. Injectables hold a significant share in hospital settings due to their use in acute care and chronic disease management, while topical, inhalation, and other niche formulations serve specific therapeutic areas such as dermatology and respiratory diseases, which are important but comparatively smaller generic segments in the Thai market.
Thailand Generic Pharmaceuticals Market Competitive Landscape
The Thailand Generic Pharmaceuticals Market is characterized by a dynamic mix of regional and international players. Leading participants such as Government Pharmaceutical Organization (GPO), Thai Nakorn Patana Co., Ltd., Siam Pharmaceutical Co., Ltd., Berlin Pharmaceutical Industry Co., Ltd., Biopharm Chemicals Co., Ltd., Greater Pharma Co., Ltd., Atlantic Pharmaceutical Co., Ltd., T.O. Pharma Co., Ltd., Bio Generic Pharma Co., Ltd., Mega Lifesciences Public Co., Ltd. (Generics Portfolio), Bangkok Lab & Cosmetic Co., Ltd. (Pharma Division), Biolab Co., Ltd., S.M.P. Pharmaceutical Co., Ltd., Sriprasit Pharma Co., Ltd., General Drugs House Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space.
Thailand Generic Pharmaceuticals Market Industry Analysis
Growth Drivers
- Increasing Demand for Affordable Medications:The Thai government reported that approximately 60% of the population relies on generic medications due to their affordability. In future, the average cost of generic drugs is projected to be 30% lower than branded alternatives, making them accessible to a broader demographic. This demand is further fueled by rising healthcare costs, which reached THB 1.3 trillion, prompting consumers to seek cost-effective solutions for their medical needs.
- Government Initiatives to Promote Generic Drugs:The Thai government has implemented various policies to encourage the use of generic drugs, including the establishment of the National Health Security Office (NHSO) which allocated THB 220 billion for generic drug procurement in future. These initiatives aim to enhance public health by ensuring that essential medications are available at lower prices, thereby increasing the market share of generics in the pharmaceutical sector, which is currently at 42%.
- Rising Healthcare Expenditure:Thailand's healthcare expenditure is projected to reach THB 1.6 trillion in future, reflecting a 7% increase from the previous period. This growth is driven by an expanding healthcare infrastructure and increased investment in public health initiatives. As healthcare spending rises, the demand for affordable generic medications is expected to grow, as consumers and healthcare providers seek cost-effective alternatives to manage rising medical costs effectively.
Market Challenges
- Intense Competition from Local and International Players:The Thai generic pharmaceuticals market is characterized by fierce competition, with over 120 local manufacturers and numerous international companies vying for market share. In future, the market is expected to see a 17% increase in new entrants, intensifying price wars and potentially squeezing profit margins for existing players. This competitive landscape poses significant challenges for companies aiming to differentiate their products and maintain profitability.
- Regulatory Hurdles and Compliance Issues:Navigating the regulatory landscape in Thailand can be complex, with stringent requirements for drug registration and approval. In future, the Thai Food and Drug Administration (FDA) is expected to process over 1,600 applications, leading to potential delays in market entry for new generic drugs. Compliance with evolving regulations can also incur significant costs, impacting the ability of companies to launch products efficiently and effectively.
Thailand Generic Pharmaceuticals Market Future Outlook
The future of the Thailand generic pharmaceuticals market appears promising, driven by increasing healthcare demands and supportive government policies. As the population ages and healthcare expenditures rise, the market is likely to see a shift towards more affordable medication options. Additionally, advancements in technology and drug development are expected to enhance the efficiency of generic drug production, further solidifying the role of generics in the healthcare system. The focus on sustainability will also shape future practices in the industry.
Market Opportunities
- Expansion into Emerging Therapeutic Areas:There is significant potential for growth in emerging therapeutic areas such as oncology and chronic disease management. With the Thai government investing THB 55 billion in research and development for these sectors in future, generic manufacturers can capitalize on this trend by developing affordable alternatives to expensive treatments, thereby increasing their market presence.
- Collaborations with Healthcare Providers:Forming strategic partnerships with healthcare providers can enhance market access for generic drugs. In future, hospitals and clinics are expected to increase their procurement of generics by 22%, driven by cost-saving initiatives. Collaborations can facilitate better distribution channels and improve awareness of generic options among healthcare professionals, ultimately benefiting both parties.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Drug Type | Branded Generics Unbranded (INN) Generics Biosimilar Generics Specialty / Complex Generics Others |
| By Formulation | Oral Solids (Tablets, Capsules) Oral Liquids (Syrups, Suspensions) Injectables Topical (Creams, Ointments, Gels) Inhalers & Nasal Preparations Others |
| By Therapeutic Area | Cardiovascular Anti-diabetes Anti-infectives Central Nervous System Oncology Respiratory Gastrointestinal Others |
| By Distribution Channel | Hospital Pharmacies (Public) Hospital Pharmacies (Private) Retail Pharmacies / Drug Stores Online Pharmacies Wholesalers / Distributors Others |
| By Route of Administration | Oral Parenteral Topical Inhalation Others |
| By Prescription Type | Prescription (Rx) Generics Over-the-Counter (OTC) Generics |
| By Manufacturer Type | Government / Public Manufacturers (e.g., GPO) Local Private Manufacturers Multinational Subsidiaries Contract Manufacturing Organizations Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration Thailand, Ministry of Public Health)
Manufacturers and Producers
Distributors and Retailers
Pharmaceutical Wholesalers
Healthcare Providers and Institutions
Industry Associations (e.g., Thai Pharmaceutical Manufacturers Association)
Financial Institutions
Players Mentioned in the Report:
Government Pharmaceutical Organization (GPO)
Thai Nakorn Patana Co., Ltd.
Siam Pharmaceutical Co., Ltd.
Berlin Pharmaceutical Industry Co., Ltd.
Biopharm Chemicals Co., Ltd.
Greater Pharma Co., Ltd.
Atlantic Pharmaceutical Co., Ltd.
T.O. Pharma Co., Ltd.
Bio Generic Pharma Co., Ltd.
Mega Lifesciences Public Co., Ltd. (Generics Portfolio)
Bangkok Lab & Cosmetic Co., Ltd. (Pharma Division)
Biolab Co., Ltd.
S.M.P. Pharmaceutical Co., Ltd.
Sriprasit Pharma Co., Ltd.
General Drugs House Co., Ltd.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Thailand Generic Pharmaceuticals Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Thailand Generic Pharmaceuticals Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Thailand Generic Pharmaceuticals Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Affordable Medications
3.1.2 Government Initiatives to Promote Generic Drugs
3.1.3 Rising Healthcare Expenditure
3.1.4 Expanding Aging Population
3.2 Market Challenges
3.2.1 Intense Competition from Local and International Players
3.2.2 Regulatory Hurdles and Compliance Issues
3.2.3 Price Sensitivity Among Consumers
3.2.4 Limited Awareness of Generic Options
3.3 Market Opportunities
3.3.1 Expansion into Emerging Therapeutic Areas
3.3.2 Collaborations with Healthcare Providers
3.3.3 Increasing Export Potential
3.3.4 Technological Advancements in Drug Development
3.4 Market Trends
3.4.1 Shift Towards Personalized Medicine
3.4.2 Growth of E-Pharmacies
3.4.3 Rising Popularity of Biologics and Biosimilars
3.4.4 Focus on Sustainable and Eco-Friendly Practices
3.5 Government Regulation
3.5.1 Drug Registration and Approval Processes
3.5.2 Pricing Regulations for Generic Drugs
3.5.3 Quality Control Standards
3.5.4 Intellectual Property Rights Enforcement
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Thailand Generic Pharmaceuticals Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Thailand Generic Pharmaceuticals Market Segmentation
8.1 By Drug Type
8.1.1 Branded Generics
8.1.2 Unbranded (INN) Generics
8.1.3 Biosimilar Generics
8.1.4 Specialty / Complex Generics
8.1.5 Others
8.2 By Formulation
8.2.1 Oral Solids (Tablets, Capsules)
8.2.2 Oral Liquids (Syrups, Suspensions)
8.2.3 Injectables
8.2.4 Topical (Creams, Ointments, Gels)
8.2.5 Inhalers & Nasal Preparations
8.2.6 Others
8.3 By Therapeutic Area
8.3.1 Cardiovascular
8.3.2 Anti-diabetes
8.3.3 Anti-infectives
8.3.4 Central Nervous System
8.3.5 Oncology
8.3.6 Respiratory
8.3.7 Gastrointestinal
8.3.8 Others
8.4 By Distribution Channel
8.4.1 Hospital Pharmacies (Public)
8.4.2 Hospital Pharmacies (Private)
8.4.3 Retail Pharmacies / Drug Stores
8.4.4 Online Pharmacies
8.4.5 Wholesalers / Distributors
8.4.6 Others
8.5 By Route of Administration
8.5.1 Oral
8.5.2 Parenteral
8.5.3 Topical
8.5.4 Inhalation
8.5.5 Others
8.6 By Prescription Type
8.6.1 Prescription (Rx) Generics
8.6.2 Over-the-Counter (OTC) Generics
8.7 By Manufacturer Type
8.7.1 Government / Public Manufacturers (e.g., GPO)
8.7.2 Local Private Manufacturers
8.7.3 Multinational Subsidiaries
8.7.4 Contract Manufacturing Organizations
8.7.5 Others
9. Thailand Generic Pharmaceuticals Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Revenue Band & Employee Count)
9.2.3 Thailand Generic Revenue (USD / THB, Latest Year)
9.2.4 3-year CAGR in Generic Revenues
9.2.5 EBITDA Margin from Generics Portfolio
9.2.6 Market Share in Thai Generics (%)
9.2.7 Product Portfolio Breadth (No. of Generic SKUs / Molecules)
9.2.8 Share of Hospital vs Retail Channel Sales
9.2.9 R&D Spend as % of Sales
9.2.10 Capex as % of Sales (Manufacturing & Capacity Expansion)
9.2.11 Export Revenue Share from Thailand Operations
9.2.12 Average Price Discount vs Originator Brand (%)
9.2.13 Tender Win Rate in Public Procurement (%)
9.2.14 Working Capital Days (Receivables + Inventory)
9.2.15 Compliance / Quality Track Record (Regulatory Flags, GMP Certifications)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Government Pharmaceutical Organization (GPO)
9.5.2 Thai Nakorn Patana Co., Ltd.
9.5.3 Siam Pharmaceutical Co., Ltd.
9.5.4 Berlin Pharmaceutical Industry Co., Ltd.
9.5.5 Biopharm Chemicals Co., Ltd.
9.5.6 Greater Pharma Co., Ltd.
9.5.7 Atlantic Pharmaceutical Co., Ltd.
9.5.8 T.O. Pharma Co., Ltd.
9.5.9 Bio Generic Pharma Co., Ltd.
9.5.10 Mega Lifesciences Public Co., Ltd. (Generics Portfolio)
9.5.11 Bangkok Lab & Cosmetic Co., Ltd. (Pharma Division)
9.5.12 Biolab Co., Ltd.
9.5.13 S.M.P. Pharmaceutical Co., Ltd.
9.5.14 Sriprasit Pharma Co., Ltd.
9.5.15 General Drugs House Co., Ltd.
10. Thailand Generic Pharmaceuticals Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Public Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Education
10.1.4 Ministry of Defense
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Energy Efficiency Initiatives
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Pharmacies
10.3.3 Clinics
10.4 User Readiness for Adoption
10.4.1 Awareness of Generic Options
10.4.2 Training and Support Needs
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Cost Savings Analysis
10.5.2 Expansion into New Therapeutic Areas
11. Thailand Generic Pharmaceuticals Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from the Thai Food and Drug Administration (FDA)
- Review of industry publications and white papers on generic pharmaceuticals
- Examination of trade statistics and import/export data from the Ministry of Commerce
Primary Research
- Interviews with key opinion leaders in the pharmaceutical sector, including pharmacists and healthcare professionals
- Surveys conducted with executives from leading generic pharmaceutical companies
- Focus group discussions with patients and healthcare providers to understand perceptions of generic drugs
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including government reports and industry surveys
- Triangulation of qualitative insights from interviews with quantitative data from market reports
- Sanity checks through expert panel reviews comprising industry veterans and academic researchers
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total pharmaceutical market size in Thailand and the share attributed to generics
- Analysis of healthcare expenditure trends and their impact on generic drug consumption
- Incorporation of demographic data to assess potential market growth segments
Bottom-up Modeling
- Collection of sales data from major generic pharmaceutical manufacturers operating in Thailand
- Estimation of market share based on product categories and therapeutic areas
- Volume and pricing analysis to derive revenue projections for the generic segment
Forecasting & Scenario Analysis
- Development of forecasting models based on historical growth rates and market trends
- Scenario analysis considering regulatory changes and shifts in consumer behavior towards generics
- Projections for market growth under various economic conditions through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmacy Retailers | 120 | Pharmacy Owners, Store Managers |
| Healthcare Professionals | 100 | Doctors, Nurses, Pharmacists |
| Generic Drug Manufacturers | 80 | CEOs, Product Managers |
| Patients Using Generic Medications | 100 | Chronic Disease Patients, General Consumers |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Policy Makers |
Frequently Asked Questions
What is the current value of the Thailand Generic Pharmaceuticals Market?
The Thailand Generic Pharmaceuticals Market is valued at approximately USD 2.0 billion, representing a significant portion of the overall pharmaceutical market, which is estimated at around USD 8.9 billion. This growth is driven by increasing demand for affordable medications and government initiatives promoting generics.